Fabrication of Nasicon strong electrolyte-based Na-air battery
Using ambient air as a gas supply, we constructed Na-air cells primarily based on Nasicon strong electrolyte (SE), incorporating an air-electrode that includes Nasicon (Na3Zr2Si2PO12) SE as an ionic conductor and nickel (Ni) steel as a substitute of carbon because the digital conductor (Fig. 1a–d). This design will be realized by the superior chemical stability of Nasicon SE in ambient air20. Earlier studies finding out Na-air batteries with hybrid electrolytes (aqueous and SE) clearly show that the facet reactions between Nasicon and the opposite parts (H2O, O2, and CO2) aren’t noticed, and dependable electrochemical efficiency was doable although dense Nasicon SE was in direct contact with H2O in addition to flowing gases comparable to O2 and CO221,22. Furthermore, the employment of strong electrolytes may help suppress the chemical reactions between the air and the Na steel anode and using the carbon-free air-electrode can mitigate unpredictable facet reactions, generally induced by carbon (Supplementary Desk S1)14. Dense Nasicon SE was synthesized by a solid-state response and had ionic conductivity of ~0.2 mS cm−1 at room temperature (RT) (Supplementary Fig. S1). To scale back the interfacial resistance between dense strong electrolyte and the air-electrode, and enhance the exercise within the air-electrode, a duplex strong electrolyte that’s composed of the dense strong electrolyte and a porous strong electrolyte was ready. The dense strong electrolyte was firstly ready after which the porous Nasicon layer was fabricated on the dense Nasicon by a screen-printing course of utilizing a Nasicon slurry with a pore former (Di-ethylene glycol butyl ether). After this, the duplex (porous + dense) strong electrolyte (Fig. 1a, b) was heated at 1100 °C to sinter the Nasicon slurry and improve contact between porous and dense Nasicon SE. For electron conduction, Ni nanoparticles had been shaped contained in the porous Nasicon of the duplex construction by an infiltration course of utilizing Ni(NO3)2 · 6H2O aqueous answer (Fig. 1b). Lastly, a porous Ni present collector was laminated on the porous Nasicon with infiltrated Ni nanoparticles by the screen-printing course of with Ni slurry containing the pore former (Fig. 1a). The Ni present collector was chosen as a result of it has comparatively excessive electrochemical stability with water in comparison with different metals; no reactions or modifications in chemical nature of the air-cathode (Nasicon + Ni steel) had been noticed even after linear sweep check (Supplementary Fig. S2). The ensuing assemblage (Fig. 1a, d) had ~50 μm thickness of the air-electrode, comprised of porous Nasicon, Ni nanoparticle, and porous Ni layer as a present collector.

a Schematic diagram of the Nasicon SE-based Na-air cell with carbon-free air-electrode and the duplex strong electrolyte. Microstructures of the SE-based Na-air cell: (b) cross-sectional and (c) prime view of the air-electrode. Optical photos of (d) air-electrode and (e) anode facet of the cell.
To scale back interfacial resistance on the anode facet, a skinny gold (Au) layer (thickness ~10 nm) was deposited between Na steel and the dense Nasicon SE (Fig. 1a, e) utilizing an ion coater. The interfacial resistance considerably decreased (Supplementary Fig. S3) as a result of the formation of Na-Au alloy could make a homogeneous contact on the interface23,24,25,26. The cell was assembled (Supplementary Fig. S4), after which its air-electrode facet was purged with O2 fuel (99.995% purity) for 3 h earlier than electrochemical check to extend the cell’s integrity (Supplementary Fig. S5). The purged O2 can induce the formation of a safety layer that may defend the Na steel anode. We speculate that ambient air, particularly moisture, can cross over to the Na anode facet by pores in dense strong electrolyte or by non-perfect sealing of the cell, general inflicting extreme contamination of Na steel.
Understanding the electrochemical redox mechanism of SE-based Na-air battery
The Nasicon SE-based Na-air cell was electrochemically cycled for 10 cycles in open air with 70% relative humidity (RH) at 25 °C (Fig. 2a). The present densities had been 0.02 mA cm−2 on the 1st cycle, and elevated to 0.1 mA cm−2 in subsequent cycles. This means that ambient air can certainly be used as a gas for reversibly working the Na-air cell. It must be famous that the cell exhibits greater electrochemical exercise in ambient air than in different gases with out moisture (O2, CO2, N2, and their mixtures) (Supplementary Fig. S6a) implying the significance of moisture in activating reversible electrochemical response with the ambient air. Within the 1st cycle, the discharge proceeds by electrochemical response with a voltage plateau at ~2.5 V, whereas the cost course of exhibits a response with a voltage plateau at ~3.6 V. As cycle proceeds, extra voltage plateaus at ~3.2 and ~3.4 V within the discharge seem (dQ/dV curve in Fig. 2a) after which start to extend in capability. It was additionally confirmed that the redox potentials noticed in cyclic voltammetry measurement are in line with the voltage profile within the fixed present situation (Supplementary Fig. S6b). After 10 cycles, the plateaus at 3.2 V and three.4 V are virtually saturated with out additional rising the capability. This means that the electrochemical reactions in subsequent cycles have modified from the first cycle within the Na-air cell with the ambient air.
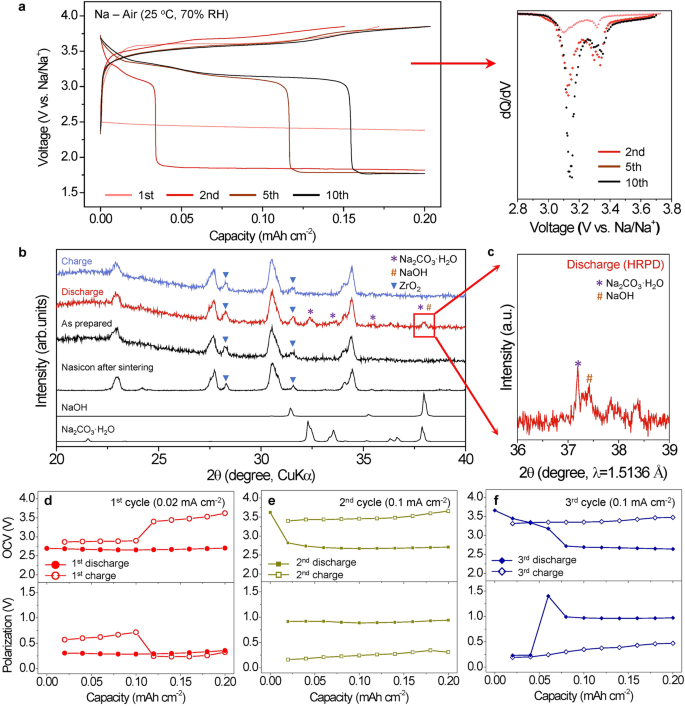
a Voltage profiles (left) and differential discharge capability plots, dQ/dV for the 2nd to tenth cycles (proper) throughout the first 10 cycles below the ambient air situation with cutoffs set at 0.2 mAh cm−2 for discharge and three.85 V for cost. The present densities had been set at 0.02 mA cm−2 for the primary cycle and 0.1 mA cm−2 for the following cycles. b Ex-situ XRD patterns of the air-electrodes in a pristine state, after 1st discharge, and after 1st cost. (Voltage profiles: Supplementary Fig. S7). c Ex-situ synchrotron X-ray powder diffraction of the discharged air-electrode after the first cycle. d–f OCVs of the cell (higher) and its polarization (decrease) from the GITT measurements throughout (d) 1st cycle, (e) 2nd cycle, and (f) third cycle. Present pulses of 0.02 mA cm−2 (1st cycle) and 0.1 mA cm−2 (2nd and third cycles) for 12 min, adopted by a relaxation for 1 h after every pulse had been employed. All information in Fig. 2 had been obtained by testing cells in open air with 70% RH at 25 °C.
To characterize the response merchandise from the first cycle, ex-situ X-ray diffraction (XRD) measurements had been carried out on the 2 air-electrodes (Fig. 2b). One electrode was discharged to 0.8 mAh cm−2 and the opposite was charged as much as 3.85 V simply after the discharge (Supplementary Fig. S7). The XRD clearly exhibits that Na2CO3·H2O and NaOH within the discharged electrode are shaped after which their content material diminishes attributable to electrochemical decomposition throughout cost. Synchrotron excessive decision X-ray powder diffraction (Fig. 2c) additional confirms the co-existence of NaOH and Na2CO3·H2O within the discharged electrode by observing the separation of the height at ~37° in XRD. Ex-situ Raman spectra of the air-electrodes after discharge and cost in 1st cycle additional confirms that the formation and decomposition of Na2CO3·H2O (x = 0 or 1) within the SE-based Na-air cell (Supplementary Fig. S8). In-situ Differential electrochemical mass spectrometry (DEMS) evaluation was additionally carried out to verify decomposition of Na2CO3·H2O (Supplementary Fig. S9). In-situ DEMS clearly exhibits {that a} CO2 fuel is developed throughout cost. Contemplating fuel evolution price primarily based on the response, among the developed CO2 is saved within the air-electrode (particularly within the absorbed H2O).
To take a better have a look at the electrochemical reactions upon biking within the Nasicon SE-based Na-air cell, galvanostatic intermittent titration approach (GITT) measurements had been carried out at 25 °C below open air of 70% RH for 3 cycles (Fig. 2nd–f). The utilized present density was 0.02 mA cm-2 throughout the 1st cycle and elevated to 0.1 mA cm−2 throughout the 2nd and third cycles. All currents had been utilized for 10 pulses with durations of 1 h throughout the 1st cycle and with durations of 12 min throughout the 2nd and third cycles, adopted by a relaxation for 1 h after every pulse. Open circuit voltages (OCVs) of the Na-air cell within the 1st cycle (Fig. 2nd) had been fairly totally different from these within the subsequent cycles. This clearly signifies that totally different electrochemical redox reactions happen within the 1st cycle in comparison with the following cycles, and the discharge merchandise can range throughout biking. Within the 1st discharge, a single discharge voltage plateau seems at ~2.7 V, whereas there are two primary voltage plateaus throughout the 1st cost: one at ~2.7 V, which will be the corresponding response of the noticed discharge redox response, and one other at ~3.4 V, which can be a further redox response. These two distinct voltages within the 1st cost counsel that two Na compounds can kind as response merchandise throughout or after the first discharge. On condition that each Na2CO3ˑH2O and NaOH within the discharged electrode are noticed in XRD (Fig. 2b, c) and Raman spectra (Supplementary Fig. S8), and solely a single electrochemical redox response at ~ 2.7 V within the discharge is noticed within the 1st discharge, solely one of many two Na compounds is shaped electrochemically throughout the 1st discharge whereas the opposite just isn’t. Contemplating the thermodynamic redox potential of NaOH (Supplementary Desk S2) and the comparatively excessive focus of O2 and H2O in ambient air, NaOH may very well be electrochemically shaped at ~2.7 V throughout the 1st discharge (Eqs. (1) and (2)). The next response describes the origin of the one voltage plateau noticed.
On the Na electrode facet:
$${{{{{rm{Na}}}}}}leftrightarrow {{{{{{rm{Na}}}}}}}^{+}+{{{{{{rm{e}}}}}}}^{-}$$
(1)
On the air-electrode facet:
$${{{{{{rm{Na}}}}}}}^{+}+{{{{{{rm{e}}}}}}}^{-}+0.5{{{{{rm{H}}}}}}_2{{{{{rm{O}}}}}}({{{{{rm{g}}}}}})+0.25{{{{{rm{O}}}}}}_2({{{{{rm{g}}}}}})leftrightarrow {{{{{rm{NaOH}}}}}}({{{{{rm{s}}}}}}),{{{{{rm{E}}}}}}=2.75{{{{{rm{Vvs}}}}}}.{{{{{rm{Na}}}}}}/{{{{{{rm{Na}}}}}}}^{+}$$
(2)
Noting that two Na compounds had been noticed within the 1st discharge and sodium carbonates have greater thermodynamic stability than NaOH (Supplementary Desk S2), the formation of Na2CO3·H2O could also be attributable to chemical reactions of NaOH with CO2 and H2O from the ambient air throughout or after the first discharge by following the under reactions (Eqs. (3) and (4)):
$$2{{{{{rm{NaOH}}}}}}+{{{{{{rm{CO}}}}}}}_{2}to {{{{{{rm{Na}}}}}}}_{2}{{{{{{rm{CO}}}}}}}_{3}+{{{{{{rm{H}}}}}}}_{2}{{{{{rm{O}}}}}}$$
(3)
$$2{{{{{rm{NaOH}}}}}}+{{{{{{rm{CO}}}}}}}_{2}to {{{{{{rm{Na}}}}}}}_{2}{{{{{{rm{CO}}}}}}}_{3}{{cdot }}{{{{{{rm{H}}}}}}}_{2}{{{{{rm{O}}}}}}$$
(4)
Due to this fact, the 2 plateaus noticed within the 1st cost (Fig. 2nd) will be ascribed to the electrochemical decomposition of NaOH at ~2.7 V (Eqs. (1) and (2) and Na2CO3·xH2O (x = 0 or 1) at ~3.4 V (Eqs. (1), (5) and (6), respectively.
On the air-electrode facet:
$${2{{{{{rm{Na}}}}}}}^{+}+{2{{{{{rm{e}}}}}}}^{-}+{{{{{{rm{CO}}}}}}}_{2}left({{{{{rm{g}}}}}}proper)+{0.5{{{{{rm{O}}}}}}}_{2}left({{{{{rm{g}}}}}}proper)leftrightarrow {{{{{{rm{Na}}}}}}}_{2}{{{{{{rm{CO}}}}}}}_{3}left({{{{{rm{s}}}}}}proper),{{{{{rm{E}}}}}}=3.37, {{{{{rm{V}}}}},{vs}}., {{{{{rm{Na}}}}}}/{{{{{{rm{Na}}}}}}}^{+}$$
(5)
$${2{{{{{rm{Na}}}}}}}^{+}+ {2{{{{{rm{e}}}}}}}^{-}+{{{{{{rm{CO}}}}}}}_{2}({{{{{rm{g}}}}}})+{0.5{{{{{rm{O}}}}}}}_{2}({{{{{rm{g}}}}}})+{{{{{{rm{H}}}}}}}_{2}{{{{{rm{O}}}}}}({{{{{rm{g}}}}}}) leftrightarrow {{{{{{rm{Na}}}}}}}_{2}{{{{{{rm{CO}}}}}}}_{3}cdot {{{{{{rm{H}}}}}}}_{2}{{{{{rm{O}}}}}}({{{{{rm{s}}}}}}),{{{{{rm{E}}}}}}=3.43, {{{{{rm{V}}}}}, {vs}}., {{{{{rm{Na}}}}}}{/{{{{{rm{Na}}}}}}}^{+}$$
(6)
The OCVs of the primary cost plateau barely will increase from ~2.7 V to ~2.9 V (Fig. 2nd). Contemplating that NaOH·H2O tends to be shaped when NaOH is uncovered in humid air27, we speculate that NaOH·H2O can be chemically shaped on the finish of discharge and it may possibly result in the rise within the OCVs of the primary cost plateau (see NaOH·H2O reactions in Supplementary Desk S2). After the first cycle, the extra redox response at ~3.4 V in subsequent discharge cycles begins to develop and turns into dominant, whereas the redox response at ~2.7 V in subsequent cost cycles disappears after the first cycle (Fig. 2e, f). Given the thermodynamic potentials and the differential capability (dQ/dV) plot (Fig. 2a), the voltage plateau at ~3.4 V is usually a results of the formation of sodium carbonates comparable to Na2CO3·xH2O (x = 0 or 1) (Eqs. (5) and (6).
To know the discharged merchandise within the SE-based Na-air cell, ex-situ measurements within the air-electrodes that had been pre-cycled for 10 cycles had been carried out. (Supplementary Fig. S10). Electrochemical formations of Na2CO3·xH2O at ~3.4 V and NaOH at ~2.0 V had been confirmed through the use of ex-situ XRD and Raman spectroscopy measurements. When the SE-based Na-air cell was discharged to 2.8 V (Supplementary Fig. S10a), the formation of the discharge merchandise (Na2CO3·xH2O) was clearly noticed by ex-situ Raman spectroscopy. Raman measurement clearly exhibits that the CO32− band of Na2CO3 and OH bands of H2O was detected within the cell discharged to 2.8 V. In-situ Raman evaluation additionally exhibits the direct formation of Na2CO3·xH2O throughout discharge (Supplementary Fig. S11). When the discharge course of was additional proceeded to 0.8 mAh cm−2 and to 1.5 mAh cm−2 at ~2.0 V, the depth of XRD peaks comparable to NaOH within the ex-situ air-electrodes repeatedly elevated in comparison with these of Na2CO3·xH2O and Nasicon. This means that the response at ~2.0 V is ascribed to the electrochemical formation of NaOH. Combining these outcomes, we will conclude that the Na2CO3·xH2O and NaOH is electrochemically shaped at excessive (~3.4 V) and low (~2.0 V) potential, respectively. The electrochemical response of Na2CO3·xH2O within the Na-air cell is activated and turns into the predominant redox response as biking proceeds. That is the primary report of a reversible response through the electrochemical formation of carbonate compounds within the Li/Na-ambient air cells. It must be famous that the electrochemical decomposition/formation of Na2CO3·xH2O (Eqs. (5) and (6)) within the Na-air cell happens at a lot greater redox potential than reported carbonate reactions11,28,29,30 partly as a result of the sodium carbonate reactions within the Na-air cell can happen immediately with none intermediate phases or reactions.
Surprisingly, the electrochemical reactions of the Na2CO3·xH2O exhibit a lot smaller polarization than that of the NaOH. Within the 1st cycle, examined at 0.02 mA cm−2, the polarizations of the NaOH redox response had been ~0.3 V for the discharge and ~0.6 V for the cost, whereas the polarization of the Na2CO3·xH2O reactions occurred at ~3.4 V was ~0.25 V for the first cost (Fig. 2nd). Additionally, even at elevated the present density of 0.1 mA cm−2 after 1st cycle (5 occasions greater than the first cycle), the polarization of the Na2CO3·xH2O decomposition/formation reactions occurred at ~3.4 V within the cost/cost in Fig. 2e, f was virtually just like that of the first cycle. This means that the electrochemical decomposition/formation reactions of Na2CO3·xH2O will be kinetically facile, in contrast to earlier results9,13. In distinction, the NaOH response exhibits sharp enhance within the polarization from roughly 0.3 V at 0.02 mA cm−2 to about 0.9 V at 0.1 mA cm−2 (Fig. 2nd, e, f). Sodium carbonate’s electrochemical reactions in Nasicon SE-based Na-air cells are considerably activated and improved by a thermodynamic response pathway throughout the charging/discharging course of. Consequently, the Na-air battery with the ambient air in subsequent cycles will ship greater redox potential (~3.4 V) than Na-O2 cell5,6, and obtain low polarization even with primary electrochemical reactions of sodium carbonates.
Results of the absorbed H2O on electrochemical reactions within the SE-based Na-air cell
To additional characterize electrochemical/chemical reactions throughout biking, we carried out scanning electron microscopy (SEM) measurements for the air-electrodes of two cells. One was discharged to 0.8 mAh cm−2 and the opposite was charged to three.85 V after the discharge (Supplementary Fig. S7). The air-electrodes of those two cells had been in contrast with the pristine cell. Earlier than the cell check, the air-electrode was noticed as a particulate kind (Fig. 3a and Supplementary Fig. S12a). Nonetheless, the discharge merchandise within the air-electrode confirmed a film-like morphology that covers your complete electrode after 1st discharge (Fig. 3b and Supplementary Fig. S12b) indicating that the electrochemical/chemical response is widespread within the electrode. Many of the film-like discharge merchandise disappeared after 1st cost. It will probably show that the discharge merchandise will be electrochemically decomposed (Fig. 3c and Supplementary Fig. S12c). Nonetheless, the morphology of particles within the charged cell continues to be barely blurred in comparison with that of the pristine cell. It is perhaps originated from a residual water as a result of the cost response just isn’t full throughout 1st cost (Supplementary Fig. S7). We additionally word that the discharge merchandise electrochemically shaped and decomposed even in tenth cycle (Supplementary Fig. S13) revealing the reversibility of the electrochemical reactions through the formation of sodium carbonates with NaOH (Eqs. (2), (5) and (6)). Moreover, the film-like morphology clearly exhibits that the reacted merchandise within the 1st discharge will be shaped by the absorption of liquid reactants comparable to H2O from the ambient air. On condition that NaOH, electrochemically shaped throughout 1st discharge, has a robust deliquescent property, it simply absorbs H2O from the ambient air till the NaOH dissolves into the absorbed H2O9 resulting in the formation of catholyte. The film-like morphology of the reacted merchandise will be the results of the in-situ shaped catholyte. The existence of H2O within the air-electrode was additional confirmed by electrochemical check (Supplementary Fig. S14a, b). Upon discharging the cell to 0.2 mAh cm−2 and subsequently charging it to exceed 3.9 V, a substantial enhance in capability was noticed round 3.9 V, which is analogous to the thermodynamic potential of a water decomposition21,22. This elevated capability means that H2O can keep inside the air-electrode even after the preliminary cost except a better voltage (exceeding roughly 3.9 V) is utilized. Such residual water can simply kind a blurred boundary between particles even after charging to <3.9 V. Removing of the film-like morphology by performing a vacuum drying of the discharged cell additional helps that the blurred particle boundaries are shaped by residual water (Supplementary Fig. S14c). Due to this fact, the formation of a catholyte by chemical response with the discharged merchandise of the SE-based Na-air battery is facilitated by the moisture in ambient air. This reuslt is kind of totally different from earlier Na-air studies, which present a deteriorated electrochemical response below the existence of moisture9.
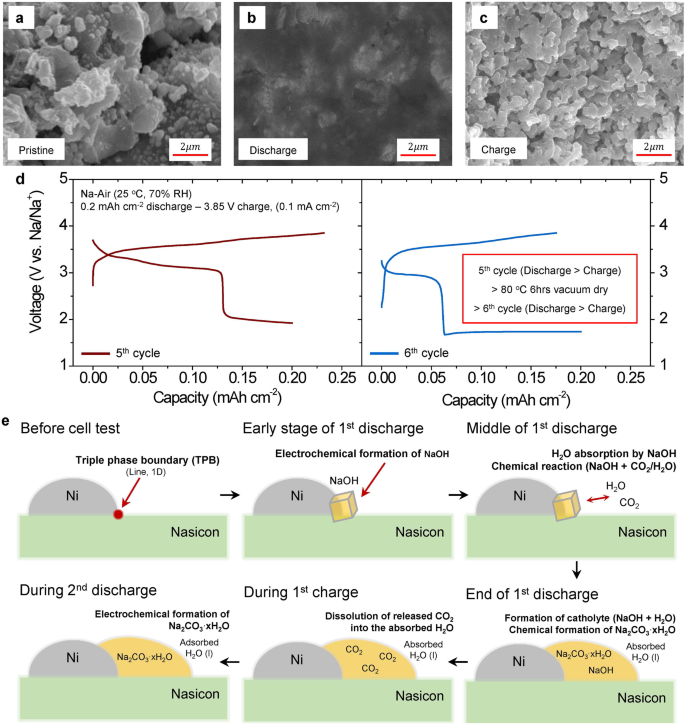
SEM photos (cross-section view) of the air-electrodes (a) within the pristine, (b) after 1st discharge (0.8 mAh cm−2), and (c) after the first cost (0.8 mAh cm−2 discharge and three.85 V cost). d Voltage profiles of fifth cycle, and sixth cycle (0.2 mAh cm−2 discharge and three.85 V cost cutoff circumstances) adopted by a vacuum drying at 80 °C for six h after fifth cost. The present density was 0.1 mA cm−2. e Schematic diagram of the activation of electrochemical Na2CO3 · xH2O reactions within the Nasicon SE-based Na-air cell with the ambient air. Knowledge in Fig. 3 had been obtained by testing the cells in open air with 70% RH at 25 °C.
The in-situ shaped catholyte concurrently facilitate each the chemical formation of Na2CO3·xH2O and the electrochemical activation of the Na2CO3·xH2O reactions. Contemplating {that a} CO2 focus within the ambient air just isn’t doable to activate the electrochemical discharge reactions of Na2CO3·xH2O, it will be affordable for considering that CO2 fuel, generated by the electrochemical decomposition of the Na2CO3·xH2O (Eqs. (5) and (6)) throughout cost, won’t be launched into the ambient air however slightly retained inside the air-electrode, significantly near the response websites. The regionally elevated retention of the CO2 fuel inside the air-electrode will be attributable to the absorbed H2O as a result of CO2 is considerably extra soluble in H2O than O231. That is additional supported by in situ Differential Electrochemical Mass Spectrometry (DEMS) evaluation, which clearly demonstrates that the precise quantity of developed CO2 throughout cost is lower than the theoretical quantity calculated by Faraday’s legislation of electrolysis (Supplementary Fig. S9). This not directly exhibits the CO2 dissolution by the absorbed H2O within the air-electrode. Consequently, the rise within the native focus of CO2 fuel within the air-electrode promotes the electrochemical formation of Na2CO3·xH2O (Eqs. (5) and (6)) within the subsequent discharge cycles. It must be famous that facet reactions associated with Ni nanoparticles (Ni(OH)2/NiOOH) is perhaps exhibited at round this potential32,33 however this impact was negligible attributable to lack of the electrochemical exercise (Supplementary Fig. S15). This means that the electrochemical reactions in strong electrolyte Na-air cell will be dominated by sodium carbonates and sodium hydroxides. Contemplating that the electrochemical formation and decomposition reactions of Na2CO3·xH2O repeatedly enhance for 10 cycles through in-situ shaped catholyte after which is saturated, the electrochemical decomposition of Na2CO3·xH2O can preserve rising the quantity of CO2 fuel contained in the air-electrode however lastly is saturated.
To know the impact of the absorbed H2O within the air-electrode on the activation of the Na2CO3·xH2O electrochemical reactions, the Na-air cell was dried up by a vacuum drying at 80 °C for six h after the cost (Fig. 3d) to take away H2O contained in the air-electrode. Earlier than performing the vacuum drying course of, the cell was cycled for 5 occasions to sufficiently activate the electrochemical reactions of Na2CO3·xH2O. The present densities had been 0.02 mA cm−2 on the 1st cycle and 0.1 mA cm−2 on the different cycles. After the activation course of, the cell on the finish of fifth cost was dried up at 80 °C for six h below vacuum situation. This vacuum drying course of severely decreased the discharge capability of the Na2CO3·xH2O from 0.13 mAh cm−2 at fifth cycle to 0.06 mAh cm−2 at sixth cycle. The consequence signifies that the lack of the absorbed H2O within the air-electrode strongly reduces the electrochemical formation of Na2CO3 · xH2O partly attributable to a lower within the quantity of the in-situ shaped catholyte, which may decrease of the CO2 focus close by the response websites contained in the air-electrode. This demonstrates that the existence of the absorbed H2O considerably impacts for the activation of the electrochemical response of Na2CO3 · xH2O.
To additional perceive the origin of the carbonate reactions, the cells had been constructed with the ‘hybrid (aqueous electrolyte with NaOH or Na2CO3 and Nasicon strong electrolyte)’ electrolyte system; it’s just like the discharged state of the Nasicon SE primarily based Na-air battery with the in-situ shaped catholyte. The cell with the hybrid electrolyte was ready by filling the aqueous answer (1 mL) to the air-cathode facet. As well as, the air-electrode facet of the cell was closed to suppress water evaporation throughout cell exams (Supplementary Fig. S16a) in contrast to the Na-air cell, which is open to the ambient air. Electrochemical exams had been carried out at RT. The ~3.4 V discharge plateau was barely elevated by biking the cell with the NaOH (aq) hybrid electrolyte (Supplementary Fig. S16b). Quite the opposite, the cell with the Na2CO3 (aq) hybrid electrolyte confirmed the numerous enhance of the ~3.4 V plateau and exhibited the same voltage curve to the Nasicon SE primarily based Na-air cell working within the ambient air (Supplementary Fig. S16c). To additional perceive the position of CO2 for the electrochemical formation/ decomposition of Na2CO3·xH2O, the SE-based Na-O2 bubbled H2O cell, which is CO2-free cell, was ready after which examined (Supplementary Fig. S16d). It didn’t present the ~3.4 V voltage response, and didn’t have the continual activation of ~3.4 V response throughout cycles in comparison with the SE-based Na-air cell. As well as, Raman measurements had been carried out to be able to observe the discharge product within the cell with Na2CO3 (aq) hybrid electrolyte. Raman spectroscopy measurements of the electrodes (Supplementary Fig. S17) present that carbonates and hydroxides had been shaped at ~3.4 V and ~2.0 V, respectively throughout discharging the cell. On condition that the electrochemical reactions in hybrid Na-air cell with Na2CO3 (aq) answer is kind of comparable with these of the SE-based Na-air cell, it not directly helps that Na2CO3 is electrochemically shaped at ~3.4 V within the SE-based Na-air cell. These outcomes clearly show that the ~3.4 V response is said to the formation of Na2CO3·xH2O and the CO2 from the air is an integral part for activating this carbonate response. Contemplating that electrochemical decomposition of Na2CO3 within the presence of water throughout cost can activate the electrochemical response at ~3.4 V within the subsequent discharge, it may be demonstrated that the carbonate reactions within the Nasicon SE-based Na-air cell will be activated by the dissolution of CO2 in H2O. In consequence, the in-situ shaped catholyte within the Nasicon SE-based Na-air cell throughout cycles critically impacts the activation of the Na2CO3·xH2O reactions and its reversible electrochemical response.
Determine 3e exhibits a schematic diagram describing the activation of electrochemical reactions with Na2CO3ˑxH2O and their reversibility within the SE primarily based Na-air cell with the ambient air. Originally of the first discharge, NaOH is shaped electrochemically by reacting with O2/H2O from the ambient air on the triple section boundary of Nasicon, Ni steel, and air (Eq. (2)). The discharge product, NaOH, spontaneously absorbs H2O from the ambient air till it’s dissolved within the absorbed H2O resulting in the formation of the catholyte. Then, the in-situ shaped catholyte (NaOH +H2O) can chemically react with CO2 within the air to yield Na2CO3 xH2O throughout and after 1st discharge (Eqs. (3) and (4)). These discharged merchandise can simply cowl your complete electrode resulting in the formation of the film-like morphology that may considerably enhance the lively response space attributable to in-situ shaped catholyte. Throughout 1st cost, the Na2CO3 xH2O and NaOH are electrochemically decomposed after which launch CO2 and O2 or H2O (Eqs. (2), (5) and (6)). If the CO2 and O2 gases are developed throughout 1st cost, the absorbed H2O would possibly seize these gases (particularly CO2) close by the response websites attributable to excessive CO2 fuel solubility in H2O31. Consequently, on the finish of cost, CO2 focus on the within the air-electrode can enhance drastically, after which within the following (2nd) discharge the electrochemical formation of Na2CO3 xH2O (Eqs. (5) and (6) will be activated. The looks of a number of peaks within the dQ/dV plot at ~3.4 V (Fig. 2a) is probably going associated not solely to the electrochemical formation of Na2CO3 but additionally the change in OCV from the Na2CO3 xH2O reactions (Eqs. (2), (5) and (6)) attributable to varied states (i.e. strong, fuel, or aqueous) of reactants/merchandise (Supplementary Desk S2). In the course of the 2nd discharge, the redox reactions eat many of the dissolved CO2 fuel within the catholyte after which subsequently, the electrochemical response of NaOH (Eq. (2)) happens on the finish of discharge. The formation of NaOH on the finish of every discharge, which corresponds to the voltage plateau under 2 V after 2nd cycle, can result in extra absorption of H2O and CO2 from the air at every cycle (Supplementary Fig. S18), which may allow to kind the catholyte and assist electrochemical decomposition of Na2CO3 xH2O in subsequent cost processes. Additionally, kinetics of the chemical reactions between NaOH and CO2 (Eqs. (3) and (4)) are additionally improved by the presence of the absorbed H2O9. This permits many of the NaOH to kind Na2CO3 xH2O on the finish of the 2nd and third discharge as noticed within the GITT check (Fig. 2e, f). Consequently, absolutely the quantity of CO2 fuel contained in the air-electrode will be elevated, ensuing within the gradual enhance of the Na2CO3 xH2O reactions in subsequent discharge cycles (Fig. 2a).
Moreover, the in-situ shaped catholyte can considerably enhance different essential electrochemical properties of the Nasicon SE-based Na-air battery. The catholyte, which will increase the lively space, allows a rise of the achievable discharge capability (Supplementary Fig. S19) by round 1.5 occasions (~6.3 mAh cm−2) when in comparison with the utmost capability calculated primarily based on the pore quantity within the air-electrode (Supplementary Desk S3). As well as, an extra quantity of the discharge product was discovered even on the Pt mesh (4 in Supplementary Fig. S4) that was on the porous Ni present collector (3 in Fig. 1a) and was not involved with any ionic conductor (Supplementary Fig. S20). This consequence signifies that the in-situ shaped catholyte can act as a brand new ionic conductor for rising the lively response space.
Electrochemical performances of the SE-based Na-air cells in ambient air
The SE-based Na-air cell exhibits glorious electrochemical performances together with superior capability retention and excessive price functionality below open air circumstances with 70% RH (Fig. 4a–c) and 40% RH (Fig. 4d–f) as a result of in-situ shaped catholyte regardless of working by the electrochemical response of carbonates and hydroxides.
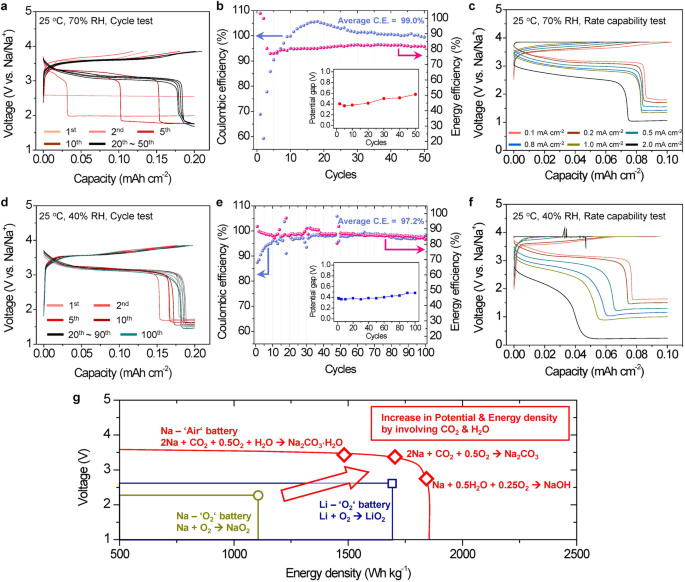
a–c Cycle and Fee functionality exams in open air with 70% RH at 25 °C: (a) Voltage profile and (b) efficiencies of the cycle check (inset: potential hole of the carbonates response between cost and discharge calculated from the dQ/dV plots) at 0.1 mA cm−2 inside discharge and cost cutoffs of 0.2 mAh cm−2 and three.85 V, respectively. c Fee functionality check carried out with discharge and cost cutoffs set at 0.2 mAh cm−2 and three.85 V, respectively, using a 3.85 V CCCV mode till the present diminished to lower than 30% of its preliminary worth. d–f Cycle and Fee functionality exams in open air with 40% RH at 25 °C, below the identical present density and cutoff circumstances as detailed in a–c: d Voltage profile and e efficiencies of the cycle check (inset: potential hole of the carbonates response between cost and discharge calculated from the dQ/dV plots). Prior to those exams, the cell underwent a pre-activation course of for 10 cycles at 70% RH and 25 °C (Fig. 2a). f Fee functionality check following the activation course of (Supplementary Fig. S21a). The legends in f are similar to these in c. g Potential-Vitality density plot evaluating the metal-O2 cells primarily based on superoxides with the SE-based Na-air cells.
Within the cycle exams, the cells had been discharged to 0.2 mAh cm−2 and charged to three.85 V. The present densities had been 0.02 mA cm−2 at 1st cycle and 0.1 mA cm−2 in subsequent cycles. When the cell was examined in ambient air with 70% RH, the voltage plateau at ~3.4 V (comparable to the Na2CO3·xH2O reactions) prolonged with rising cycle quantity. This extension was saturated on the tenth cycle with the capability of the plateau reaching ~0.16 mAh cm−2 and the vitality effectivity (=Edischarge/Echarge) converging to ~80% (Fig. 4a, b). It must be famous that the NaOH response on the finish of discharge cycles nonetheless seems indicating the formation of the catholyte through the delinquency of NaOH will be sustained (Supplementary Fig. S18). After the activation of the carbonate reactions within the first 10 cycles, the cell examined in 70% RH confirmed glorious cycle stability. The cell confirmed 99.0% common coulombic effectivity and 82.1% common vitality effectivity for 50 cycles. The coulombic effectivity between tenth and twenty fifth cycle is greater than 100% (Fig. 4b), which can be attributed to residual discharge merchandise forming between the first and tenth cycles the place the effectivity is decrease than 100%. After the activation course of for 10 cycles, the kinetics of the Na-air cell are improved, permitting the residual discharge merchandise to decompose throughout tenth~twenty fifth cycles and thus the coulombic effectivity will be elevated. The vitality effectivity is far greater than the opposite metal-air cells in earlier studies3,6,8,11,13 as a result of the electrochemical response pathways throughout cost and discharge are the identical (Na2CO3 · xH2O reactions) within the Na-air cell. Excessive vitality effectivity will be achieved because the potential hole of the Na2CO3 xH2O reactions between cost and discharge, ~0.4 V, is small (Fig. 4b). This illustrates that the Na-air cell operated in ambient air will be reversibly cycled through reversible Na2CO3 xH2O reactions and may enhance vitality effectivity.
The speed functionality check was carried out by discharging the cells with the present densities from 0.1 mA cm−2 to 2.0 mA cm−2 and charging the cells utilizing a continuing present – fixed voltage (CCCV) protocol: charging to three.85 V at fixed present, after which making use of a voltage maintain at 3.85 V till the present reaches <30% of the utilized present (Fig. 4c). The CCCV methodology was carried out to completely cost the cell with out the decomposition of water above ~3.9 V (Supplementary Fig. S14).
Earlier than the speed functionality check, the cell was pre-activated for 10 cycles in ambient air with 70% RH (Supplementary Fig. S21a) to make sure that the Na2CO3 xH2O response was absolutely saturated. In the course of the price functionality check, the cell was cycled 5 occasions at every present density (Supplementary Fig. S21b); solely the voltage profile of the final cycle at every present density is proven (Fig. 4c). The cell may very well be operated at a excessive present density of two.0 mA cm−2 although an elevated polarization was noticed. At 2.0 mA cm−2 the coulombic effectivity was ~88%, and the vitality effectivity was ~66%. This consequence clearly demonstrates that the electrochemical reactions involving carbonates and hydroxides within the Nasicon SE primarily based Na-air cell are kinetically facile, partly as a result of in-situ shaped catholyte by chemical response of the discharged merchandise with the air.
The cycle and price functionality exams had been additionally carried out in air with decreased RH, from 70% to 40% (Fig. 4d–f), to be able to perceive the impact of RH on the electrochemical efficiency. When the cell was cycled in air with solely 40% RH, the Na2CO3 xH2O reactions had been barely activated and elevated even with repeated cycles (Supplementary Fig. S22a), and thereby the polarization was a lot greater than the cell in air with 70% RH. This means that the quantity of H2O strongly impacts the electrochemical activation of the carbonate reactions initially cycles. To activate the electrochemical reactions of Na2CO3 · xH2O in ambient air with 40% RH, the SE-based Na-air cell was pre-cycled in air with 70% RH for 10 cycles (Fig. 2a and Supplementary Fig. S21a). After pre-cycling the cell in air with 70% RH, the cycle and price functionality exams in air with 40% RH had been carried out. The electrochemical properties of the cells in air with 40% RH (Fig. 4d) had been similar to these in air with 70% RH (Fig. 4a). This consequence implies that when the ample quantity of the catholyte is shaped and the sodium carbonate response is activated within the air-electrode below the air with excessive RH, the reversible Na2CO3 xH2O response is effectively maintained even in air with low RH in subsequent cycles. Surprisingly, the cell with 40% RH confirmed considerably improved cycle stability in comparison with the cell with 70% RH (Fig. 4e); the cell with 40% RH air confirmed secure capability retention to 100 cycles with 97.2% common coulombic effectivity and 86.5 % common vitality effectivity. Additionally, the potential hole between the cost and discharge throughout the cycle check (~0.4 V) was just like that of the cell in air with 70% RH (Fig. 4e). Lengthy prolonged cycle stability signifies that the reversible electrochemical reactions of Na2CO3 xH2O will be sustained even with much less quantity of H2O in air if the cell is absolutely activated by pre-cycling in air with excessive RH.
Moreover, the cell in air with 40% RH additionally confirmed affordable price functionality (Fig. 4f and Supplementary Fig. S22b). The cell was cycled as much as a excessive present density of two.0 mA cm−2 although it precipitated greater polarization than the cell with 70% RH. Additionally, the portion of the Na2CO3 xH2O reactions throughout the discharge decreased extra quickly as the present density elevated, in comparison with the cell in air with 70% RH. We additional confirmed that the SE-based Na-air cells after 10 cycles for the activation will be operated below ambient air with low RH from 20% to 40%. It was clearly noticed that the cell performances (vitality/coulombic efficiencies) decreased by reducing RH (Supplementary Fig. S23).
The cells with 70% RH confirmed the degradation within the capability retention after ~fiftieth cycles (Fig. 4b). When the Na steel was changed by new steel, the cell was recovered and confirmed a typical voltage curve of the Na2CO3 xH2O response (Supplementary Fig. S24). This consequence strongly means that the degradation will be primarily originated from the corrosion of the Na steel slightly than the air-electrode. Because the cycle lifetime of the cells was considerably prolonged by working them in air with low RH = 40% (Fig. 4e), so we speculate that inflow of air (particularly H2O in it) in cycles into the Na steel anode would possibly severely degrade the Na steel.
The electrochemical performances of the Nasicon SE-based Na-air batteries in ambient air as a gas are superior than these of all-solid-state Li-air (O2) batteries even with none liquid components in air-electrodes7,16,18, due to the upper redox potential of the Na2CO3 xH2O reactions (~3.4 V) and far smaller potential hole between the cost and discharge, which is originated from the identical electrochemical response pathway within the cost/discharge. The substantial enchancment in electrochemical efficiency within the Nasicon SE-based Na-air battery originates from the in-situ shaped catholyte that may result in the chemical reactions of the discharge merchandise shaped in 1st discharge with the ambient air, after which can activate the reversible electrochemical reactions of Na2CO3 xH2O (Fig. 4) in subsequent cycles. In consequence, these superior electrochemical performances demonstrates that the SE-based Na-air battery will be operated in ambient air with a variety of humidity by exploiting reversible electrochemical reactions of sodium carbonates/hydroxides which may ship excessive vitality density with facile kinetic.
Moreover, the carbonate reactions with excessive working voltage and a low polarization can ship greater theoretical vitality density than that of MO2 (M = Li or Na) (Fig. 4g and Supplementary Desk S4) resulting in small potential hole between cost and discharge than M2O2 or M2O (Supplementary Desk S5). In comparison with reported hybrid (aqueous + strong) electrolyte programs which have a considerable amount of H2O, the Nasicon SE primarily based Na-air battery is kind of totally different as a result of it exploits solely the absorbed H2O obtained from the humidity of the ambient air, which may present a really restricted quantity of H2O, and thereby can have a lot greater volumetric and gravimetric vitality density than the hybrid electrolyte programs. Furthermore, the reversible electrochemical reactions involving Na2CO3 · xH2O throughout the charging and discharging cycles noticed within the SE-based Na-air cell have by no means been beforehand reported within the hybrid electrolyte programs with flowing the air (not pure O2) into the aqueous electrolyte20,21,34. This may be attributable to partly due to the absorption of a minimal quantity of water that may facilitate the upkeep of a excessive native CO2 focus, which prompts the reversible Na2CO3 · xH2O reactions solely within the SE-based Na-air battery.
Using ambient air with none extra gadgets has a number of benefits for sensible use of metal-air batteries. Firstly, the cell design will be simplified as a result of extra gadgets comparable to fuel selective gadgets and fuel tanks for storing purified fuel aren’t necessary6,35. Consequently, will probably be useful enhance the gravimetric/volumetric vitality densities of the metal-air batteries and step towards into the sensible functions. Secondly, the Na-air cell in ambient air can obtain the bottom vitality price amongst varied vitality storage systems36 as a result of sodium is an earth-abundant materials, the ambient air is free, and extra price for getting ready purified fuel just isn’t obligatory. Through the use of oxide-based strong electrolytes, the Na-air battery permits to make use of ambient air reversibly as a gas, and allows to have chemical reactions between discharge merchandise and the air that may result in the formation of catholyte and may activate reversible electrochemical carbonate reactions, in distinction to earlier approaches that attempt to suppress these chemical reactions.
In conclusion, we had been capable of make use of ambient air as a gas in a Nasicon SE-based Na-air cell able to delivering reversible capability by activating electrochemical reactions with carbonates and hydroxides, that are generally believed to degrade the reversibility and induce excessive polarization in reported metal-air batteries. This counter-intuitive consequence will be defined by contemplating the position of an in-situ shaped catholyte attributable to the chemical response of the discharge merchandise with H2O, which prompts the reversible electrochemical response of carbonates and facilitate its kinetics. Consequently, the Nasicon SE-based Na-air cell delivers excessive vitality density attributable to excessive redox potential of the carbonate response in addition to excessive vitality effectivity attributable to low polarization by working on the identical electrochemical pathway throughout cost and discharge. This primary demonstration of ambient air as a gas in a SE-based Na-air battery gives steps towards creating past typical metal-O2 batteries and demonstrates the idea of using reversible carbonate reactions to allow novel electrochemical vitality platforms with strong electrolytes that may obtain a lot greater vitality density than typical batteries.








