Electrochemical efficiency of alkaline NMF//NTP coin cells
Ready NMF, NTP and business Ni/C powders have been subjected to X-ray diffraction (XRD, Supplementary Figs. 1–3), evidencing good crystallinity for functions in batteries. The influence of salt concentrations in electrolytes on HER was established by way of in-situ differential electrochemical mass spectrometry (DEMS) in NaClO4 electrolytes with chosen salt concentrations. Findings verify that, with out forming a dependable SEI, rising electrolyte focus doesn’t change the onset potential for HER (Supplementary Fig. 4).
Importantly, rising the alkalinity of electrolyte suppresses HER (Supplementary Fig. 5a-c) that contributes to decrease over-discharge (attributable to HER) of NTP anode (Supplementary Fig. 6a–c). Nonetheless, the elevated alkalinity of electrolyte drives OER (Supplementary Fig. 5d) and will increase dissolution fee of Fe and Mn elements12 of the NMF cathode, resulting in poor biking stability (Supplementary Fig. 6d, e). Compared, following coating of the Ni/C layer on the NMF electrode (thickness: ca. 1 µm, Supplementary Fig. 7), biking stability is considerably boosted (Supplementary Fig. 6f), as confirmed by way of unchanged electrolyte color (Supplementary Fig. 6g–i) along with the suppressed Fe dissolving focus within the electrolyte evidenced by way of inductively coupled plasma mass spectrometry (ICP-MS) (Supplementary Fig. 8).
The efficiency of NMF//NTP full cells utilizing a impartial electrolyte or a alkaline electrolyte with/with out Ni/C coating have been examined in a large charging voltage vary of 0.5 to 2.2 V. The NMF//NTP full cell with Ni/C coating displays a extremely boosted fee efficiency and better common discharge voltage than these with out Ni/C coating, contributing to the fast-charge means and excessive common voltage of the battery (Supplementary Figs. 9a, b). Determine 1b compares the biking performances of NMF//NTP full cells beneath three totally different circumstances at 1 C. Batteries with out Ni/C coating in each impartial and alkaline electrolytes displays a fast capability decay with capability retention of <60% following 200 cycles, whereas the alkaline-based battery with Ni/C coating displays a larger retention of ca. 100%. Considerably, the electrolyte exhibited a decrease freezing level compared with reported extremely concentrated electrolytes5,11, permitting the battery to operate at low temperatures (Supplementary Fig. 10). The battery with Ni/C coating subsequently displays a capability retention of 91.3% after 200 cycles at 0.5 C beneath −30 °C (Fig. 1c). Importantly, this full cell displays a document lifespan of >13,000 cycles with a excessive capability retention of 74.3% at 10 C (Fig. 1d) in alkaline electrolyte, surpassing reported performances of many aqueous batteries13.
Pouch cell efficiency and comparability
To simulate business necessities for large-scale power storage, a Ni/C coated NMF//alkaline electrolyte//NTP pouch cell was assembled with an electrode loading of ca. 20 mg cm−2. This pouch cell displays a excessive capability retention of 85% following 1,000 cycles at 500 mA g−1 (Fig. 2a). Moreover, the mass loading of the only electrode could be elevated to >30 mg cm−2 due to the low viscosity of the alkaline electrolyte of 6.0 mPa·s (Supplementary Desk 1). With this excessive loading, NMF//NTP pouch cell displays steady biking life with a capability retention of ca. 100% inside 200 cycles at 300 mA g−1 (Fig. 2b). This massive-size pouch cell moreover exhibits excessive stability beneath ‘harsh’ circumstances of reducing and immersion in water (Supplementary Motion pictures 1–2, Supplementary Fig. 11a–c and Fig. 2c). Considerably, the minimize pouch cell powers repeatedly a digital hygrometer thermometer in water for >20 h (Supplementary Film 3, Supplementary Fig. 11d–f and Fig. second). This discovering confirms that the battery is proof against electrolyte leakage and might face up to important harm within the high-humidity surroundings. The cycled pouch cell displays no obvious quantity adjustments, evidencing that there isn’t any important fuel evolution throughout biking (Supplementary Fig. 12). The battery subsequently exhibited excessive security (low threat) for sensible software in power storage and underwater electrical gear. Importantly, the batteries exhibited a excessive biking stability and discharge capability beneath a low optimistic/adverse capability ratio of 1.06. Beneath such a low capability ratio, and 0.5 C charging fee, a excessive power density amongst ASIBs of 88.9 Wh kg−1 is achieved (Fig. 2e and Supplementary Desk 2). The cell-level power density of the brand new ASIB primarily based on a digital cell configuration with real looking parameters was computed, Supplementary Desk 3. The compacted density and porosity of the electrode, the quantity of electrolyte, measurement of the present collector, tab and package deal, have been decided utilizing empirical parameters derived from the reported literature assuming a 10-layer pouch cell geometry. The power density was computed by way of dividing the whole power by the whole mass of the pouch cell. The anticipated battery power density is ca. 61 Wh kg−1. In contrast with reported electrochemical storage gadgets, this new battery displays important benefits together with, use of ample components (equivalent to Fe, Mn and Ti), excessive security (excessive tolerance of excessive humidity surroundings), environmental benignity (non-poisonous electrolyte) and an extended lifespan (Fig. 2f and Supplementary Desk 4).
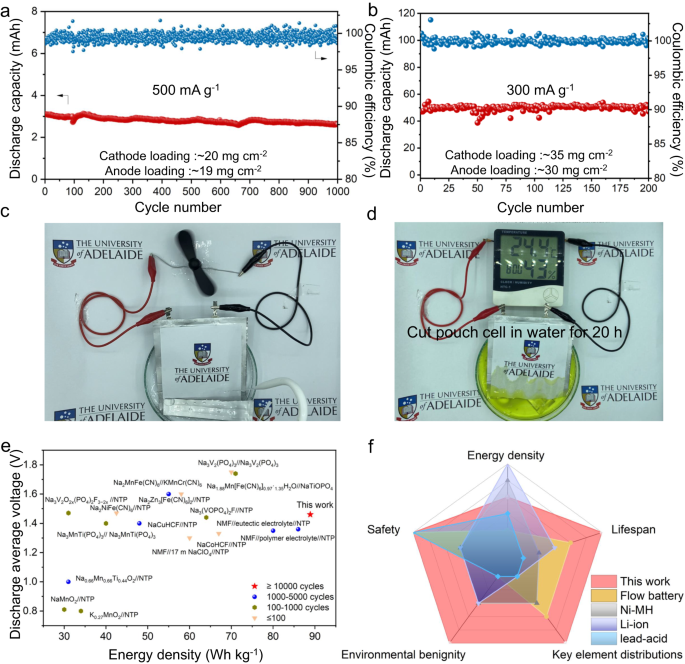
a Biking efficiency for NMF//NTP pouch cell at a present density of 500 mA g−1. b Biking efficiency for NMF//NTP pouch cell at 300 mA g−1. c Digital {photograph} of a minimize pouch cell to energy a fan in water. d Digital {photograph} of a minimize pouch cell to energy a humidity clock in water. e Comparability of lifespan and power density with reported ASIBs. f Comparability of current work with business batteries (as quantified in Supplementary Desk 4).
Origin of H3O+-rich native surroundings
To find out the underlying components for top efficiency of the alkaline ASIB, the interface construction with Ni/C coating was assessed by way of in-situ ATR-IR spectroscopy. The carbon coating was taken as a management group to eradicate any influences of carbon and Nafion-Na assist. For the electrode modified by pure carbon, the spectra exhibited no obvious change regardless of being charged to 1.3 V (vs. Ag/AgCl), evidencing that the carbon and assist don’t change native surroundings of the cathode floor (Fig. 3a, the operate of carbon in Ni/C coating is mentioned in Supplementary Textual content S1 and Supplementary Fig. 13). In distinction, with Ni/C modification, new peaks at 1798 and 2032 cm−1 seem when the potential is >0.6 V, attributed to 2 uneven O-H stretching modes of H3O+ (({{{{{{rm{nu }}}}}}}_{{{{{{{rm{H}}}}}}}_{3}{{{{{{rm{O}}}}}}}^{+}}^{{{{{{{rm{a}}}}}}}^{1}}) and ({{{{{{rm{nu }}}}}}}_{{{{{{{rm{H}}}}}}}_{3}{{{{{{rm{O}}}}}}}^{+}}^{{{{{{{rm{a}}}}}}}^{2}})). Peaks for the resonance state for the uneven O-H stretching modes in H3O+ (({nu }_{{H}_{3}{O}^{+}}^{{a}^{2},,r+u})) at 2223 cm−1 and the umbrella vibration for H3O+ (({nu }_{{H}_{3}{O}^{+}}^{u})) at 1230 cm−1 are additionally visible14,15 confirming that the H3O+ accumulation on the electrode floor is induced by Ni nanoparticles. Operando DEMS was used to research water decomposition on this alkaline battery throughout biking. The battery with out Ni/C coating displays HER and OER concurrently at a low optimistic/adverse capability ratio within the impartial electrolyte (Supplementary Fig. 14). Nonetheless, following coating Ni/C on the NMF cathode in alkaline electrolyte, each HER and OER usually are not obvious, aside from hint O2 on the first cycle earlier than activating the floor coating (Fig. 3b). It’s concluded subsequently that the H3O+-rich native surroundings induced by Ni/C protecting layer suppresses OER within the alkaline electrolyte, while the alkaline electrolyte retards HER.
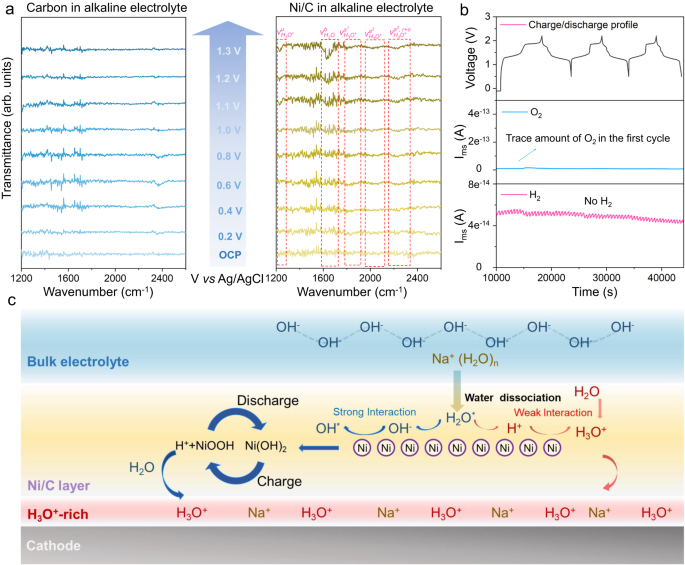
a ATR-FTIR spectra for pure carbon and Ni/C at totally different potentials. Elementary excitations of H3O+, denoted ({{{{{{rm{nu }}}}}}}_{{{{{{{rm{H}}}}}}}_{3}{{{{{{rm{O}}}}}}}^{+}}^{{{{{{rm{u}}}}}}}) (umbrella vibration), ({{{{{{rm{nu }}}}}}}_{{{{{{{rm{H}}}}}}}_{3}{{{{{{rm{O}}}}}}}^{+}}^{{{{{{{rm{a}}}}}}}^{1}}) (uneven O-H stretching), and resonance states between elementary excitation of uneven O-H stretching and mixture tones (r + u) denoted ({{{{{{rm{nu }}}}}}}_{{{{{{{rm{H}}}}}}}_{3}{{{{{{rm{O}}}}}}}^{+}}^{{{{{{{rm{a}}}}}}}^{2},,{{{{{rm{r}}}}}}+{{{{{rm{u}}}}}}}), the place r and u signify, respectively, pissed off rotation and umbrella vibration. Bending of H2O is denoted ({{{{{{rm{nu }}}}}}}_{{{{{{{rm{H}}}}}}}_{2}{{{{{{rm{O}}}}}}}^{}}^{{{{{{rm{b}}}}}}}). b Operando DEMS findings to find out H2 and O2 evolution throughout NMF//NTP battery biking at 0.5 to 2.2 V at 0.5 C. c Schematic for H3O+ accumulation mechanism on electrode floor coated with Ni/C within the alkaline electrolyte.
The H+ accumulation mechanism on the electrode floor with Ni/C is illustrated in Fig. 3c. OH− ions exhibit larger adsorption power on the Ni floor in contrast with H+ with an utilized optimistic voltage, confirmed by way of density useful principle (DFT) simulation (Supplementary Desk 5). This results in a localized lower in OH− focus beneath the protecting layer throughout charging. Along with the elevated voltage, Ni step by step undergoes oxidation, remodeling into Ni(OH)2 (Supplementary Fig. 15a, b). This additional consumes native OH−, inflicting a subsequent lower in pH. The opposite part within the coating layer, the Nafion-based polymer that is called the cationic membrane, inhibits the diffusion of OH− from the majority electrolyte to the electrode floor (Supplementary dialogue Textual content S2 and Supplementary Figs. 16–17). Subsequently, the decreased pH surroundings is maintained on the cathode floor. Ni(OH)2 subsequently converts to NiOOH (Supplementary Fig. 15b–d) which generates further H+ ions. These H+ ions generated in the course of the response stay un-neutralized due to the decreased native pH, resulting in a H3O+-rich native surroundings. The Ni(OH)2/NiOOH transformation is extremely reversible (the capability supplied by Ni/C was computed, Supplementary Fig. 18), as confirmed by way of XRPD and comfortable X-ray absorption spectra (Supplementary Fig. 19 and Supplementary dialogue Textual content S3), leading to a dynamic equilibrium increase the steadiness of H3O+-rich native surroundings beneath excessive voltages.
Ni-substitution
Along with elevated OER, the improved alkalinity of electrolyte compromises the biking stability of PBA-based cathode materials (with out Ni/C coating). Subsequently, along with inducing H3O+-rich surroundings, the Ni/C coating additionally ends in the substitution of Ni to fill Mn vacancies, thereby stabilizing the PBA cathode. The working precept for this technique is illustrated in Fig. 4a. Throughout charging, the floor of the Mn-based PBA cathode displays Mn dissolution, resulting in the era of Mn vacancies16. Within the unprotected system, the continual dissolution of Mn ions results in structural collapse and adversarial results on the cyclic stability of the battery (Fig. 4b, c). Nonetheless, within the Ni/C protected system, the in-situ substituted Ni atoms steadiness tiny structural disturbances attributable to Mn dissolution as evidenced by the steady discharge plateau of NMF//NTP battery following the coating10,17,18 (Fig. 4d). The oxidation of Ni throughout discharge produces Ni2+ ions. These Ni2+ ions step by step enter the crystalline framework to fill the Mn vacancies by forming Ni-N bonds on biking. DFT computations (Supplementary Fig. 20) for Ni substitution have been carried out to find out a ∆E worth of −8.06 eV, evidencing that Ni substitution is spontaneous.
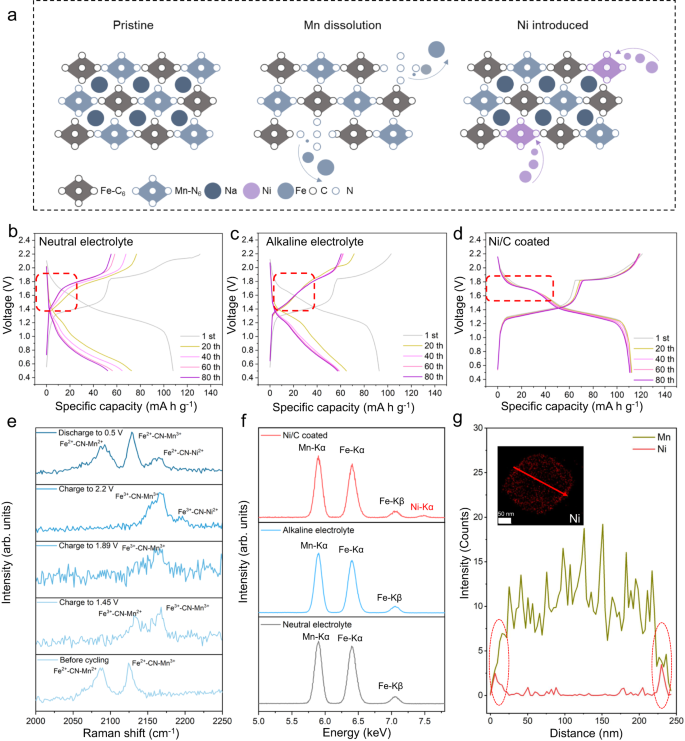
a Schematic for Ni/C coating for mitigating structural instability due to Mn dissolution in NMF cathode. Cost/discharge curves for NMF//NTP cells in (b) Impartial electrolyte (c) Alkaline electrolyte and (d) Alkaline electrolyte with Ni/C coating. e Operando Raman spectra for Ni/C coated NMF cathode cycled in alkaline electrolyte. f STEM-EDS spectra taken from NMF electrodes following biking in impartial, alkaline electrolyte and alkaline electrolyte with Ni/C coating. g STEM line scan for cycled NMF cathodes with Ni/C coating. Inset exhibits Ni mapping for NMF.
Ni substitution was additional confirmed by way of operando Raman spectra for Ni/C coated NMF cathodes cycled in alkaline electrolyte (Fig. 4e). Previous to biking, two peaks at 2089 and 2124 cm−1 are obvious, comparable to Fe2+−CN−Mn2+ and Fe2+−CN−Mn3+ vibrations, respectively19. Following charging to 1.89 V each peaks disappear, evidencing the transformation of Fe2+ to Fe3+ and Mn2+ to Mn3+. Importantly, following charging to 2.2 V, a brand new weak peak seems at 2195 cm−1 comparable to Fe3+−CN−Ni2+ 20. This discovering confirms that the introduction of Ni atoms in NMF particles follows the transformation of Mn2+ to Mn3+. Following discharging to 0.5 V, peaks for Fe2+−CN−Mn2+ and Fe2+−CN−Mn3+ shift to 2092 and 2128 cm−1 and a brand new peak seems at 2164 cm−1 that’s assigned to Fe2+−CN−Ni2+ 20, confirming the introduction of Ni.
The introduction of Ni was additionally confirmed by way of STEM-EDS mapping (Fig. 4f). There’s a new peak belonging to Ni component within the spectrum. Moreover, the EDS line scan spectra for a single NMF particle verify that Ni atoms are launched into the sting of particles to suppress the dissolution of internal Mn atoms (Fig. 4g). The STEM-energy-dispersive spectroscopy (STEM-EDS) mappings for NMF cathode with Ni/C coating following 1st, fifth and twentieth cycles (Supplementary Desk 6 and Supplementary Fig. 21) proof that the content material of Ni in NMF particles is steady after the primary cycle, confirming that the introduction of Ni into NMF cathode reaches an equilibrium within the first cycle to provide long-term stability to the battery.
To display the cathode construction stability following Ni/C safety, different characterizations have been performed. Digital images of the PBA electrodes following biking exhibits that the unprotected electrodes exhibit important steel ion dissolution in each impartial and alkaline circumstances (Supplementary Fig. 22). Compared, the dissolution phenomenon is considerably mitigated when the Ni/C protecting coating is utilized. TEM analyses proof that the construction of the PBA cathode undergoes important structural harm following biking within the alkaline situation (Supplementary Fig. 23). STEM-EDS confirms that the electrode displays Mn dissolution within the impartial media, while the concurrent dissolution of each Fe and Mn happens in alkaline electrolyte (Supplementary Fig. 24). Nonetheless, following software of the protecting coating, the electrode dissolution is considerably suppressed ensuing from in-situ Ni substitution.
The boosted structural stability of NMF cathode following introduction of inert Ni atoms in alkaline electrolyte was confirmed by way of XRPD patterns throughout cost/discharge. The structural evolutions of NMF within the first cycle is proven in Fig. 5a and Fig. 5b presents the 2D contour map for NMF reflection. The extremely reversible construction evolution throughout cost and discharge could be simply noticed in these figures. Moreover, the Rietveld refinements of NMF with/with out Ni/C coating after 1st cycle proof that each electrodes exhibit cubic phases with Fm-3m area group and a = b = c (Fig. 5c, d and Supplementary Desk 7). The lattice parameters for Ni/C coated NMF (5.28161 Å) are larger than that for uncoated NMF (5.26358 Å). This discovering is attributed to the introduction of Ni in NMF throughout biking. A rise in a (b, c) contributes to a boosted fee efficiency for the cathode, which, considerably, is in step with our findings. Importantly, in contrast with the deteriorated construction of uncoated NMF after 1st and third cycles (Fig. 5e), the overlapping patterns for Ni/C coated NMF following 1st and third cycles verify that the superb stability of NMF and Ni introduction happens at 1st cycle, as a result of in any other case, steady Ni introduction will change the XRPD sample (Fig. 5f).
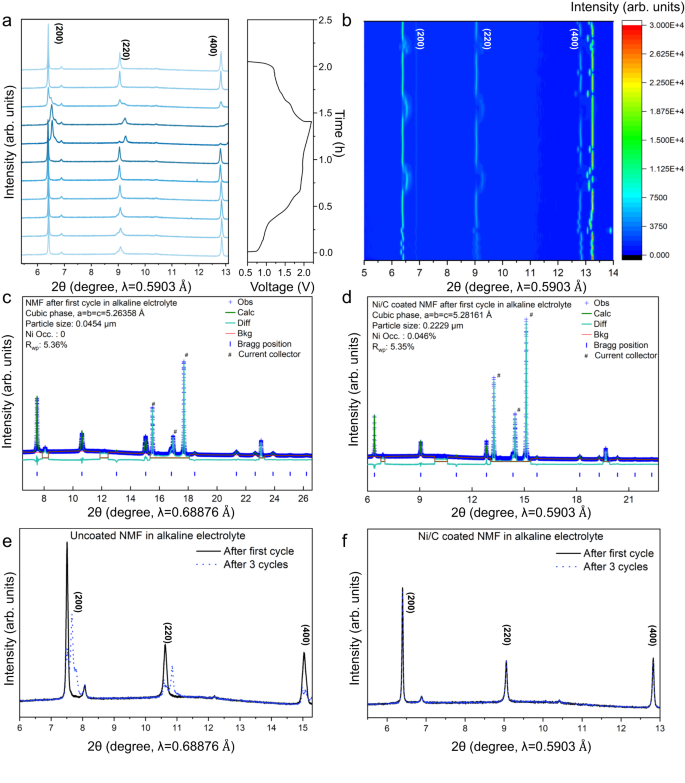
a (200), (220) and (400) reflections of synchrotron operando XRPD sample. b 2D contour plot for operando XRPD for the primary 3 cycles. Structural and Rietveld refinements for NMF cathode following 1st cycle in (c) Alkaline electrolyte and (d) Alkaline electrolyte with Ni/C coating. Comparability of XRPD sample following 1st cycle and third cycle in (e) alkaline electrolyte and (f) alkaline electrolyte with Ni/C coating.
To evaluate the doable universality of the brand new electrode modification technique in alkaline batteries, the Co/C nanoparticle was used to construct the cathode coating. Comparable with Ni nanoparticles, Co could be oxidized to Co(OH)2 in alkaline media and, it displays a reversible redox pair of Co(OH)2/CoOOH, along with the power to in-situ substitute the Mn atom. Consequently, good stability of the battery with Co/C coating is achieved (Supplementary Fig. 25). This discovering supplies proof for the universality of making H3O+-rich cathode surfaces and in-situ optimizing the NMF construction by constructing steel nanoparticle coating to spice up the efficiency of Mn-based PBA cathode in an alkaline surroundings.








