Theoretical exploration of the V2CTx MXene as an anode materials
To establish the opportunity of utilizing V2CTx MXene as a high-performance anode for AIBs, we have now carried out a collection of theoretical investigations primarily based on DFT calculations. In accordance with the earlier report, the work operate Φ which evaluates the vitality required to activate an electron from the Fermi stage to hoover was proposed to be straight associated to the electrochemical oxidation/discount potential24,25, thereby dominating the working potential. Within the number of electrodes, giant work purposeful supplies typically possess a large working potential window to extend cell performance26. Due to this fact, we give attention to the work operate of a collection of two-dimensional supplies, together with V2C (trigonal), VO2 (hexagonal)27, VS2 (hexagonal)28, V2O5 (orthorhombic)20, and V2CTx MXene (Fig. 1a) and 1T-MoS2. The plane-averaged electrostatic potential curves of V2CTx (T = O, F, OH) are displayed in Fig. 1b–c and Supplementary Fig. 1. The values of labor operate had been discovered to differ with floor terminations, decided to be 6.62 eV for V2CO2, 5.40 eV for V2CF2 and 1.85 eV for V2C(OH)2, respectively. Contemplating that the -OH termination was unlikely to adsorb the NH4+, the work operate of V2CTx (T comprises O and F) was deduced to be within the vary of 5.40 eV~6.62 eV. The work operate of 1T-MoS2 was decided to be 5.61 eV in Fig. 1d. As a consequence, the connection between the work operate and dealing potential window of investigated fashions is displayed in Fig. 1e, which signifies that the V2CTx MXene possesses the biggest work operate and widest working potential window.
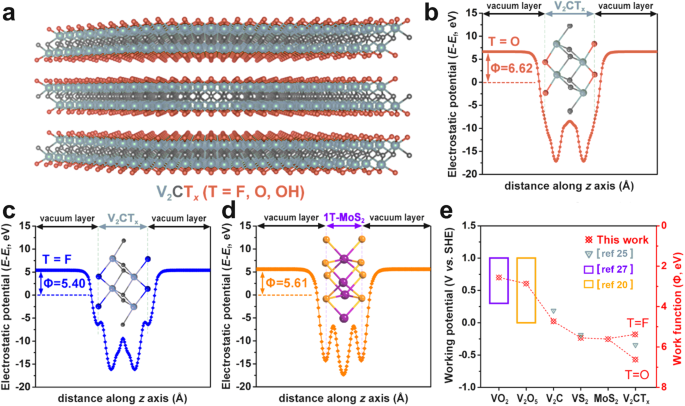
a The facet view of the construction mannequin for V2CTx MXene. Gray, black, and orange spheres denote V, C, and T terminations, respectively. b, c Aircraft-averaged electrostatic potential curve of V2CO2 and V2CF2, respectively. d Aircraft-averaged electrostatic potential curve of 1T-MoS2. e Relationship between the work operate and the working potential window of two-dimensional supplies investigated on this work.
Ammonium ion storage efficiency of V2CTx MXene
On the premise of the above theoretical calculations, it’s rational that V2CTx is predicted to own the bottom working potential window in ammonium ion storage. Then, the V2AlC bulk precursor was ready by pressureless sintering technique28. The multilayered-V2CTx (m-V2CTx) was synthesized utilizing the normal LiF + HCl etching method29. Thereafter, the delaminated-V2CTx (d-V2CTx) was obtained utilizing tetraethyl ammonium hydroxide (TBAOH) delamination therapy. All merchandise are characterised utilizing X-ray Diffraction (XRD), and the collected patterns are displayed in Fig. 2a, displaying a typical MXene construction of a layered hexagonal section. Furthermore, the diffraction peak positioned on the low-angle (2θ < 10°) vary originates from the diffraction of the (0002) crystal aircraft. Accordingly, a slight interlayer distance improve from 11.6 Å of m-V2CTx to 12.0 Å of d-V2CTx will be deduced after the TBAOH delamination therapy. The morphology of m-V2CTx MXene was then noticed utilizing transmission electron microscopy (TEM) and scanning electron microscopy (SEM), displaying a consultant accordion-like block as given in Supplementary Fig. 2. The d-V2CTx was discovered to be semitransparent below TEM remark in Fig. 2b, implying ultrathin construction with two-dimensional layer morphology. The floor space of the as-prepared MXene powders was measured by the Brunauer-Emmett-Teller (BET) technique utilizing nitrogen adsorption, which demonstrated a big rise of particular floor space after the delamination course of, from 0.996 m2 g−1 for m-V2CTx to as excessive as 26.002 m2 g−1 for d-V2CTx (Supplementary Fig. 3a). The excessive particular floor space is mostly believed to favor the publicity lively websites for ion adsorption or response. As well as, the micropores (measurement of which is smaller than 20 nm) obtained in the course of the etching course of performed an necessary function in ion transportation by way of the sheet-like d-V2CTx (Supplementary Fig. 3b). Clear lattice fringe in Fig. 2b was noticed within the high-resolution clear electron microscopy (HRTEM) picture, and a spacing of 0.255 nm corresponded carefully to the (2(bar{1}bar{1})0) planes. Chosen space electron diffraction (SAED) sample of d-V2CTx in Fig. 2c exhibited hexagonally organized sharp diffraction spots which will be listed to the [0001] zone axis sample. The factor mapping in Supplementary Fig. 4 illustrated a uniform distribution of composition components together with V, F, O, and C within the d-V2CTx nanosheet, respectively. The X-ray photoelectron spectra (XPS) check additional verified the presence of V, F, O, and C components within the d-V2CTx nanosheet (Supplementary Fig. 5a). Particularly, there exited three pairs of peaks within the high-resolution spectrum of V factor: the height positioned at 517.3 eV belonged to the V4+-2p3/2, whereas these positioned at 515.8 eV and 514.3 eV had been assigned to V3+-2p3/2 and V2+-2p3/2, respectively (Supplementary Fig. 5b). This end result indicated the co-existence of V4+, V3+, V2+ within the achieved d-V2CTx MXene.
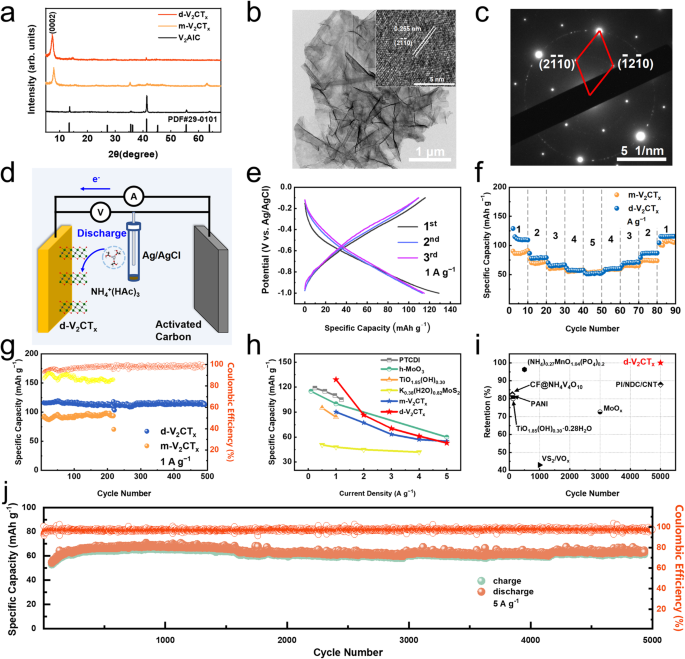
a XRD patterns of V2AlC, m-V2CTx, and d-V2CTx. b TEM and HRTEM morphology of d-V2CTx. c Collected SAED patterns of d-V2CTx. d Scheme illustrating the construction of the three-electrode half-cell. e The first-Third GCD voltage profiles. f Fee functionality. g Biking efficiency at 1 A g−1. h Lengthy-term biking efficiency at 5 A g-1 for 5000 cycles. i Fee functionality comparability with literature knowledge. j Biking efficiency comparability with beforehand reported supplies for aqueous AIBs.
Subsequently, the ammonium ion storage efficiency of V2AlC, m-V2CTx, and d-V2CTx MXene was evaluated by Swagelok-type cells with a three-electrode configuration, as proven in Fig. 2nd, with the MXene served because the working electrode, activated carbon because the counter electrode, 0.5 M NH4Ac because the electrolyte, and a saturated Ag/AgCl electrode acted because the reference electrode (E = 0.197 V vs. SHE), respectively. The galvanostatic discharge/cost (GCD) profiles in Fig. 2e demonstrated that the d-V2CTx electrode delivered a capability of 129.1 mAh g−1 at a present density of 1 A g−1 within the first discharging course of, displaying an preliminary coulombic effectivity of 89.78%. As compared, the m-V2CTx pattern exhibited a comparatively decrease particular capability of 90.2 mAh g−1 at 1 A g−1. In distinction, the pristine V2AlC pattern exhibited nearly no capability lower than 5 mAh g−1 in the identical situation (Supplementary Fig. 6), suggesting that the chemical etching strategy of V2AlC MAX was essential to attain a excessive capability.
Observe that the d-V2CTx electrode exhibited typical pseudocapacitive discharging/charging conduct displaying no obvious charging/discharging plateau in keeping with the cyclic voltammetry (CV) curve given in Supplementary Fig. 7. Furthermore, excellent price efficiency of the d-V2CTx electrode will be concluded in Fig. 2f and Supplementary Fig. 8, with a excessive particular capability of 115.9 mAh g−1 at 1 A g−1 and remaining 53 mAh g−1 at 5 A g−1. Remarkably, the speed functionality of d-V2CTx will not be solely a lot better than m-V2CTx, and likewise typical anode supplies just lately developed for ammonium ion storage together with PTCDI5, MoO39, TiO1.85(OH)0.3030, K0.38(H2O)0.82MoS231, and many others.
Lengthy-term biking stability is one other important think about evaluating the efficiency of rechargeable batteries. Determine 2g confirmed the biking performances of m-V2CTx and d-V2CTx electrodes at 1 A g−1. In a 500-cycling check, the d-V2CTx electrode delivers nearly no capability decay (114.0 mAh g−1 in 1st and 113.0 mAh g−1 in five hundredth cycle). In distinction, the m-V2CTx electrode exhibited an inferior biking efficiency (lower than 200 cycles), indicating the bigger particular floor space of d-V2CTx is useful to attain a steady particular capability. The coulombic effectivity of d-V2CTx (the orange line) remained above 95% after 500 cycles, which was clearly increased than that of m-V2CTx (the yellow line). The morphology of the d-V2CTx after 500 cycles is proven within the following SEM picture (Supplementary Fig. 9). After cycles, the fabric nonetheless maintained a layered morphology, indicating its glorious stability. The long-cycling efficiency of the d-V2CTx electrode was evaluated utilizing a excessive present density (5 A g−1) as proven in Fig. 2j. The plots point out that the capability reached its most worth after 500 cycles, and the capability retention stayed round 100% in the course of the complete 5000 cycles. To sum up, the electrochemical efficiency of the d-V2CTx electrode achieved on this work is listed in Fig. 2h, i for a quick comparability with the beforehand reported ammonium ion storage materials11,26,30,32,33, displaying passable price efficiency and superior biking stability to these recognized so far.
Determine 3a illustrates the working potential window of the d-V2CTx in addition to that of another typical ammonium ion storage supplies reported beforehand. Contemplating that little effort has been made to the examine of anode supplies for aqueous ammonium ion storage, it’s passable that the d-V2CTx possesses a comparatively low working potential vary inside −1 ~ −0.01 V (vs. Ag/AgCl), making it promising anode candidate for AIBs. Subsequently, Na0.6MnO2 (NMO) was chosen because the coupling cathode materials on this work, owing to its appropriate response potential and an ammonium ion storage capability of fifty mAh g−1 (Supplementary Fig. 10–11)34,35, thereby a full cell was constructed with the schematic two-electrode configuration as illustrated in Fig. 3b. Typical CV profiles of this d-V2CTx/0.5 M NH4Ac/NMO full cell at a present density of 1 mV s−1 delivered a pair of anodic/cathodic peaks at ≈0.6/1.0 V (Fig. 3c). GCD profiles of the as-constructed battery in Fig. 3d verified a particular capability of 170 mAh g−1 at 1 A g−1 on the first cycle (primarily based on the lively mass of the anode). The complete battery delivered a price capability of 99.1, 67.5, 56.9, 51.3, and 46.8 mAh g−1 at 1, 2, 3, 4, and 5 A g−1, respectively, and it recovered to 83.8 mAh g−1 when the present density returned again to 1 A g−1 (Fig. 3e). Long run biking recognized a biking lifetime of over 500 cycles at 1 A g−1 with a particular capability of 57.7 mAh g−1 and remained a coulombic effectivity near 100% (Fig. 3f). Our d-V2CTx/ /NMO full battery exhibited an vitality density of 103.36 Wh kg−1 at an influence density of 1127.6 W kg−1. Moreover, electrochemical Impedance Spectroscopy (EIS) was employed to additional reveal the quick ion diffusion in our system. Determine 3g depicted the Nyquist plots of our battery earlier than and after 500 cycles. From the equal circuit used to suit the EIS knowledge, the charge-transfer resistance (R2) decreased from 12.21 Ω to six.9 Ω, indicating the small barrier for cost switch inside the fabric, which was conducive to the speedy charging/discharging36. We additionally tried to confirm the sensible utility in versatile and wearable units, a soft-packed battery was constructed in Fig. 3f, which simply drove a light-emitting diode (LED) indicator below assorted bending states of 90°, demonstrating its glorious flexibility and potential utility on wearable electronics.
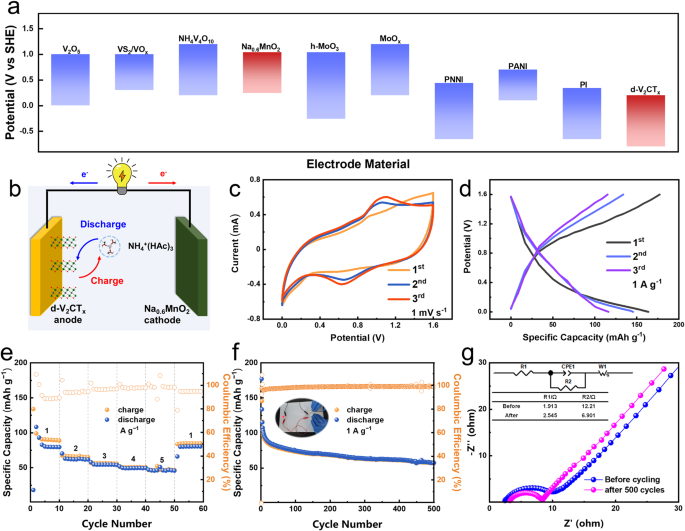
a Comparability of working potential home windows of V2CTx MXene and different ammonium ion storage supplies. b Scheme illustrating the construction of the d-V2CTx/0.5 M NH4Ac/Na0.6MnO2 full battery. c CV curves at 1 mV s−1. d GCD curves at 1 A g−1. e Fee functionality at totally different present densities of 1~5 A g−1. f Biking efficiency at 1 A g−1 with an insert displaying a soft-packed battery driving an LED mild. g EIS check earlier than and after biking.
Acetate ions enhancement impact
The d-V2CTx MXene was noticed to exhibit the characteristic of pseudocapacitance capability in the course of the storage of aqueous ammonium ions. Surprisingly, such a pseudocapacitive conduct might solely be achieved within the NH4Ac electrolyte. Determine 4a shows 5 CV curves (1 mV s−1) in varied electrolytes containing NH4+ cations. Particularly, a pair of broad redox peaks at round −0.70 V and −0.35 V was clearly noticed in 0.5 M NH4Ac electrolyte, which can’t be detected in different examined electrolytes (NH4Me, NH4Cl, (NH4)2SO4, and (NH4)2C2O4) regardless of of the identical NH4+ focus. Consequently, a a lot increased capability of 101.3 mAh g−1 was obtained in NH4Ac electrolyte than that in different electrolytes (calculated to be 80.9, 68.9, 60.3, and 52.7 mAh g−1 for (NH4)2SO4, NH4Cl, (NH4)2C2O4, and NH4Me, respectively). We additional investigated the affect of the cation species of the electrolyte salt utilizing Mg(Ac)2, Zn(Ac)2, and LiAc, displaying an obvious decline within the curve space with the absence of the redox peaks (Fig. 4b). As well as, the impact of NH4Ac on ammonium ion storage efficiency was evaluated by tailoring the salt focus. Provided that the chosen NH4+-containing aqueous electrolytes had been weakly acidic, we additional examined the electrochemical properties by pH worth regulation of the NH4Ac electrolyte. Particularly, three extra electrolytes with totally different pH values had been ready: 0.5 M HAc (pH = 1.5), 0.25 M blended resolution of HAc and NH4Ac (denoted as NH4Ac(H), pH = 3.7), and 0.25 M blended resolution of NH3·H2O and NH4Ac (denoted as NH4Ac(OH), pH = 9.8). In accordance with the CV curves proven in Fig. 4c, a slight change of the electrochemical window will be noticed, from −1 ~ −0.01 V (pH = 6.5) to −0.9 ~ −0.01 V (pH = 3.7) and additional to −0.9 ~ 0.2 V (pH = 1.5) because of the ease of hydrogen evolution in acid resolution. Nevertheless, the absence of redox response peaks yielded to a outstanding deterioration within the capability of d-V2CTx, demonstrating the significance of impartial (or almost impartial) electrolytes in facilitating redox response. The impact of NH4Ac electrolyte focus was examined lastly, as summarized in Supplementary Fig. 12. A transparent oxygen evolution response might be noticed in 10 M and 20 M NH4Ac electrolytes when charged to −0.1 V. The particular capability of d-V2CTx in 20 M NH4Ac electrolyte (88.6 mAh g−1 at 1 A g−1) was a lot inferior to that in 0.5 M NH4Ac electrolyte (115.9 mAh g−1 at 1 A g−1), suggesting {that a} excessive focus of NH4Ac electrolyte might inhibit the electrochemical efficiency of V2CTx (Supplementary Fig. 13).
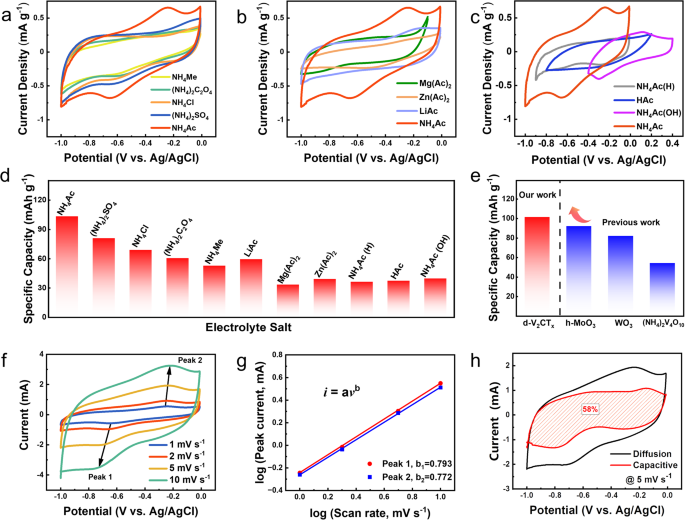
a CV curves of d-V2CTx in 0.5 M NH4+ electrolytes at 1 mV s−1. b CV curves of d-V2CTx in 0.5 M Ac− electrolytes at 1 mV s−1. c CV curves of d-V2CTx in 0.5 M NH4Ac electrolytes with pH regulation at 1 mV s−1. d Abstract of the particular capability values of d-V2CTx in several electrolytes. e Functionality comparability with different pseudocapacitive ammonium ion storage supplies. f CV curves of d-V2CTx at totally different scanning charges at 1, 2, 5, and 10 mV s−1 in 0.5 M NH4Ac displaying redox peaks. g log i versus log v plotted in keeping with redox peaks. h capacitive-controlled contribution calculated at 5 mV s−1.
To sum up, the particular capability of the d-V2CTx electrode in varied salt electrolytes is collected in Fig. 4d, displaying the optimum capability of 101.3 mAh g−1 uniquely achieved within the NH4Ac electrolyte. It’s value noting that the important thing limitation of capacitive vitality storage for each Faradaic (involving redox) and non-Faradaic (involving solely electrostatic interactions) is its low capability and unhappy vitality density. Particularly, in our case, benefited by this distinctive acetate ions enhancement impact, the particular capability surpasses the entire as-reported capacitive-typed electrodes in ammonium-ion batteries as much as date9,37,38 (Fig. 4e).
The electrochemical kinetics had been then elucidated by CV measurement at varied scanning charges at 1, 2, 5, and 10 mV s−1 in Fig. 4f. The contributions from diffusion-controlled and capacitive-controlled behaviors will be distinguished by analyzing the CV profiles in keeping with the next equation (Eq. (1)) between the height present (i) and the sweep price (v)39,40:
whereas b = 0.5 stood for a diffusion-controlled course of, and b = 1 stood for a capacitive-controlled course of. In Fig. 4g, the b worth of peak 1 and a pair of was calculated to be 0.793 and 0.772, respectively, implying that the capacitive-controlled and diffusion-controlled processes synergistically dominate the vitality storage course of. The capacitive storage contributed to ≈58% as floor pseudocapacitance at 5 mV s−1 (Fig. 4h). Moreover, the capacitive-controlled course of contributed 46%, 50%, 69% to the entire capability of d-V2CTx at 1, 2, and 10 mV s−1 (Supplementary Fig. 14). Equally, the capacitive-controlled contributions of m-V2CTx had been calculated to be 34%, 40%, 51%, 63%, respectively (Supplementary Fig. 15). These values had been decrease than these noticed in d-V2CTx, indicating an enhanced contribution from elevated floor space by delamination treatment41,42. Additionally, the capacitive storage contributions in 0.25 M (NH4)2SO4 electrolyte had been calculated utilizing the identical measurement (Supplementary Fig. 16a). The capacitive-controlled course of contributes 43%, 45%, 54%, and 69% to the entire capability of d-V2CTx at 1, 2, 5, and 10 mV s−1, respectively (Supplementary Fig. 16b). Based mostly on these knowledge, the capacitive-controlled capability was evaluated to be 46.5 mAh g−1 in 0.5 M NH4Ac electrolyte (Supplementary Fig. 17), which was increased than the 35.3 mAh g−1 in 0.25 M (NH4)2SO4 electrolyte. All the outcomes affirm {that a} distinctive pseudocapacitive response occurs within the NH4Ac electrolyte and enhances the capacitive-controlled course of.The redox response which considerably contributed to the capability of d-V2CTx in NH4Ac electrolyte was engaging, the mechanism of which might be mentioned in our following sections. The ex-situ XPS characterization and in-situ electrochemical quartz crystal microbalance (EQCM) measurement was carried out. The ex-situ XPS outcomes clearly recognized the evolution of chemical composition in addition to the valence state of d-V2CTx in the course of the Faraday course of (Supplementary Fig. 18). The lively supplies within the working electrode utilizing a three-electrode configuration after 5 discharging/charging cycles had been chosen as a consequence of its coulombic effectivity approached 100% at the moment. Within the high-resolution V 2p3/2 XPS spectrum, it was notable that the valence state of V on the absolutely charged state was composed of +4 and +3, which is totally different from the preliminary state of V2CTx (+4, +3, and +2)43. Through the discharging course of, the attribute peak of V4+ (517.7 eV) was evident at Level A (−0.1 V) however disappeared at Level B (−0.7 V). As a substitute, the height of V3+ (516.5 eV) elevated considerably, accompanied by the looks of the V2+ peak (513.9 eV). Within the charging course of, the valence state of V remained fixed at Level C (−1 V). Whereas charged to Level D (−0.15 V), the V4+ peak rose once more to a excessive stage. Moreover, O 1 s XPS spectra indicated a reversible transformation of O valence, during which the attribute peak (530.4 eV) of the V−O bond decreased at −0.7 V within the discharging course of, and recovered at −0.15 V within the charging course of. The opposite peak at 532.0 eV represented the V-O···HN which elevated at −0.7 V within the discharging course of. In any other case, ex-situ XRD was employed to look at the modifications in layer spacing in the course of the charging and discharging course of, as proven in Supplementary Fig. 19. The diffraction peak representing the V2CTx (0002) side at absolutely discharged state confirmed nearly no change in comparison with that on the absolutely charged state, suggesting that the interlayer spacing of the d-V2CTx electrode typically stayed unchanged throughout the entire electrochemical course of.
In-situ EQCM measurement gives an efficient method to understanding the deposition/dissolution course of on the electrode floor thereby disclosing the cost storage mechanism. Subsequently, a three-electrode cell using quartz microcrystals as the present collector was constructed as proven in Supplementary Fig. 20. Determine 5a presents the CV curve with a possible window of −1.0 to −0.01 V versus Ag/AgCl of the EQCM cell at 2 mV s−1. The curve gave the impression to be comparatively clean from level A to B, then one peak rose between −0.5 ~ −0.7 V in the course of the discharging course of. In accordance with Sauerbrey’s equation and Faraday equation (see in Strategies), the obvious molar mass of interacted ions (Mw, g mol−1 e−1) correlated with the mass change per coulomb ((Delta m)/(Delta Q)) which will be calculated from the slope in Fig. 5b. Accordingly, the mass elevated slowly within the vary of −0.01 V ~ −0.6 V (from level A to B), which had been estimated with a Mw of 18 g mol−1 e−1. Usually, there are two doable interactive species (NH4+, Ac−) which may be related to 0.5 M NH4Ac electrolyte. Observe that the as-detected worth of 18 g mol−1 e−1 could be very near the molecular weight of NH4+ group, such a mass improve will be attributed to the adsorption of NH4+ group on MXene floor. In −0.7 V ~ −1.0 V vary (from level C to D), the mass elevated till the top of the discharge interval with a Mw of 196 g mol−1 e−1, which was near the mass sum of 1 NH4+ and three HAc molecules ([NH4+(HAc)3]). Conversely, there occurred a two-stage mass drop in the course of the charging course of. First, the mass decreased by 18 g mol−1 e−1 from A’ to B’, and the Mw all of the sudden modified to 196 g mol−1 e−1 from C’ to D’ when the potential reached −0.4 V. Mixed with the CV curve in Fig. 5a, the massive Mw worth of 196 g mol−1 e−1 occurred within the strategy of the redox response, making it rational to be attributed to the redox response between the electrode and electrolyte.
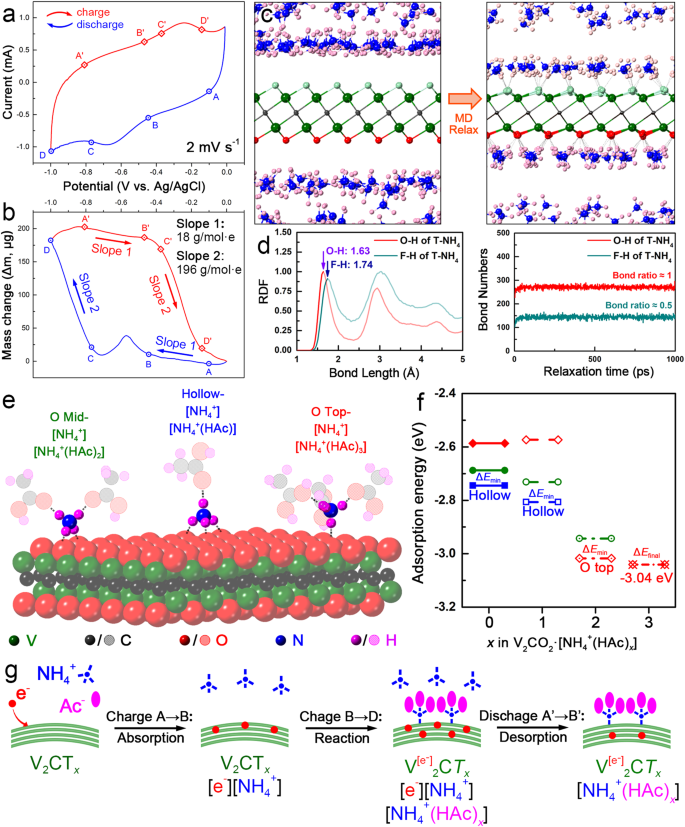
a CV curves of d-V2CTx in 0.5 M NH4Ac electrolytes within the EQCM cell. b mass change of d-V2CTx in a single discharging/charging course of. c adsorption configuration evaluation obtained by MD simulation (d) the radial distribution operate (RDF) and bond numbers between V2CTx (T = -F, -O) and [NH4+] ions; (d) adsorption configurations obtained by DFT calculation displaying the interplay between V2CO2 and [NH4+], [NH4+(HAc)x] teams. e the adsorption vitality calculation outcomes on the floor of V2CO2. f DOS evaluation of V factor in V2CO2 and V2CO2[NH4+(HAc)3]. g scheme displaying the adsorption of [NH4+] ions on V2CO2 and response of [NH4+(HAc)x] teams with V2CO2.
On this work, each DFT calculation and molecular dynamics (MD) simulation had been carried out to raised perceive the ion/electron switch and pseudocapacitive origin within the NH4Ac electrolyte. The [NH4+] ion possessing a singular tetrahedral-shaped multipole was able to rotating to take care of a subset of coordinated H-bonds with 4 oxygen atoms from each electrode and electrolyte6. After a full leisure in each DFT calculation and MD simulation, the doable adsorption configurations of [NH4+] ions and [NH4+(HAc)x] teams on the floor of V2CO2 had been collected and displayed in Fig. 5c–e, Supplementary Fig. 21–22, which will be typically divided into three varieties in keeping with the variety of hydrogen bonds: the “O top-[NH4+]” (with the central N positioned above O termination, labeled by pink) bonds with 1×O termination from V2CO2 which was able to holding 1 ~ 3×HAc; the “O mid-[NH4+]” (with the central N positioned on the center of two O terminations, labeled by inexperienced) bonds with 2×O terminations from V2CO2 which was able to greedy 1 ~ 2×HAc; the “Hole-[NH4+]” (with the central N positioned on the hole of three O terminations, labeled by blue) bonds with 3×O terminations which was able to dangling 1×HAc. Furthermore, the MD simulation in Fig. 5c–d, Supplementary Fig. 23–24 clearly demonstrated {that a} a lot increased protection price of the [NH4+] ions will be obtained on the floor of the V2CO2 mannequin than V2CF2 and V2C(OH)2. This end result signifies that V2CTx having wealthy -O terminations can obtain excessive capacitance. An in depth DFT calculation of the adsorption vitality was displayed in Fig. 5e. The Hole-[NH4+] mannequin possessed the bottom formation vitality (−2.74 eV) amongst all three doable ion adsorption configurations, which was energetically extra favorable than the fashions of O mid-[NH4+] (−2.69 eV) and O top-[NH4+] (−2.59 eV). Notably, there exists an additional Coulomb interplay between V2CTx and NH4+, from the adverse floor potential of V2CTx and the constructive cost of NH4+ which is uncared for within the DFT simulation. The truth is, the adsorption of NH4+ ions on the floor of V2CTx within the experiment scenario was extra possible than within the theoretical scenario. As a consequence, {the electrical} double-layer capability (EDLC) occurred within the first stage of discharging course of, equivalent to the 18 g mol−1 e−1 mass gaining (A → B) noticed within the EQCM check. With the saturation of [NH4+] adsorption (strongly depending on the floor space of V2CTx), the vitality storage mechanism turned to pseudocapacity (PC) the place the valence state of V modified with the incidence of response between V2CTx and [NH4+(HAc)x] teams. It has been broadly accepted that the alternation of floor terminations, typically from -O to -OH, performed a big function within the electron storage mechanism of MXene electrodes. Whereas within the aqueous ammonium ion system using a impartial (or almost impartial) NH4Ac electrolyte, the lean of proton made such a termination alternation tough. As a consequence, the response between V2CTx and [NH4+(HAc)x] facilitated the redox response, by offering -O termination with a [NH4+] which acted as a pseudo-proton. The optimum group adsorption configurations of [NH4+(HAc)x] had been achieved at Hole-[NH4+(HAc)] for 1×HAc (−2.81 eV), O top-[NH4+(HAc)2] for two×HAc (−3.02 eV) and O top-[NH4+(HAc)3] 3×HAc (−3.04 eV), respectively (Fig. 5f). The configurations of teams point out a crucial rotation of [NH4+] ions with the rising variety of HAc molecules. As a consequence, the floor response of [NH4+(HAc)x] group primarily contributed to the pseudocapacity within the second stage of discharging course of, equivalent to the 196 g mol−1 e−1 mass gaining (C → D) noticed within the EQCM check, and the rotation of [NH4+] ions as properly the alternation of floor termination of V2CTx led to a floor group redistribution which accounted for the mass fluctuation (B → C) noticed within the EQCM curve. The change within the valence state of V components posed an ideal impression on the bonding properties of V2CTx. Supplementary Fig. 25 plotted the density of states (DOS) of V-d orbital in V2CO2 and V2CO2[NH4+(HAc)3], respectively. It may be seen that the V3d-O2p hybridization primarily contributed to the orbitals round Fermi stage. With the introduction of [NH4+(HAc)3] group on the floor, basic segregation of wave-functions round Fermi stage will be noticed: the bonding orbitals (BO) under Fermi stage barely shifted in the direction of low vitality (the realm middle of which strikes from −3.60 eV to −3.64 eV), indicating a doable strengthening impact on V-O bonding, whereas the anti-bonding orbitals (ABO) above Fermi stage barely shifted in the direction of excessive vitality (the realm middle of which moved from 0.92 eV to 1.14 eV, implying a doable enhancing functionality to accommodate excited electrons, particularly with the presence of an additional orbit at 2.15 eV. The cost density distinction plotted in Supplementary Fig. 26 demonstrated that floor terminations of V2CTx performed a big function in realizing the cost switch between V factor and [NH4+(HAc)3] group: in keeping with the Bader evaluation end result, the valence state of V decreased from +1.78 (constructive for shedding electrons) to +1.69 with the presence of 1 [NH4+(HAc)3] group on the floor of V2CO2 (a 3 × 3 × 1 supercell), which align with the conclusions from ex-situ XPS characterization (Supplementary Fig. 18).
Lastly, the ammonium ion storage mechanism of the V2CTx electrode within the NH4Ac electrolyte was summarized in Fig. 5g. Within the first stage of discharging course of, the electrons had been saved on the floor of V2CTx electrode, dominated by the EDLC mechanism accompanied by the electrostatic adsorption of [NH4+] ions. Within the second stage of discharging course of, the electrons had been saved on the V website of V2CTx electrode, yielding to the alternation of valence state companied by the coordination of floor terminations (from -O to -O···HN), relating with the floor response between V2CTx and [NH4+(HAc)x] teams, which triggered the as-observed notable pseudocapacitive conduct. The entire electrochemistry response equation (Eqs. (2–5)) will be illustrated as follows:
(1)discharging course of:
$${{{{{{rm{V}}}}}}2{{{{{rm{CT}}}}}}}_{{{{{{rm{x}}}}}}}+{{{{{{{rm{NH}}}}}}}_{4}}^{!+}+{{{{{{rm{e}}}}}}}^{{{{{{rm{-}}}}}}}to {{{{{{rm{V}}}}}}2{{{{{rm{CT}}}}}}}_{{{{{{rm{x}}}}}}}{{cdot }}({{{{{{rm{e}}}}}}}^{{{{{{rm{-}}}}}}}){{cdot }}left[{{{{{{{rm{NH}}}}}}}_{4}}^{!+}right]({{{{{rm{adsorption}}}}}},, {{{{{rm{EDLC}}}}}})$$
(2)
$${{{{{{rm{V}}}}}}2{{{{{rm{CT}}}}}}}_{{{{{{rm{x}}}}}}}+{{{{{{{rm{NH}}}}}}}_{4}}^{!+}{({{{{{rm{HAc}}}}}})}_{3}+{{{{{{rm{e}}}}}}}^{{{{{{rm{-}}}}}}}to {({{{{{rm{V}}}}}},{{{{{{rm{e}}}}}}}^{{{{{{rm{-}}}}}}})}_{2}{{{{{{rm{CT}}}}}}}_{{{{{{rm{x}}}}}}}{{cdot }}left[{{{{{{{rm{NH}}}}}}}_{4}}^{!+}{({{{{{rm{HAc}}}}}})}_{3}right]({{{{{rm{discount}}}}}},, {{{{{rm{PC}}}}}})$$
(3)
(2)charging course of:
$${{{{{{rm{V}}}}}}2{{{{{rm{CT}}}}}}}_{{{{{{rm{x}}}}}}}{{cdot }}({{{{{{rm{e}}}}}}}^{{{{{{rm{-}}}}}}}){{cdot }}left[{{{{{{{rm{NH}}}}}}}_{4}}^{!+}right]to {{{{{{rm{V}}}}}}2{{{{{rm{CT}}}}}}}_{{{{{{rm{x}}}}}}}+{{{{{{{rm{NH}}}}}}}_{4}}^{!+}+{{{{{{rm{e}}}}}}}^{{{{{{rm{-}}}}}}}({{{{{rm{desorption}}}}}})$$
(4)
$$({{{{{rm{V}}}}}},, {{{{{{rm{e}}}}}}}^{{{{{{rm{-}}}}}}}){2{{{{{rm{CT}}}}}}}_{{{{{{rm{x}}}}}}}{{cdot }}left[{{{{{{rm{NH}}}}}}4}^{!+}{({{{{{rm{HAc}}}}}})}_{3}right]to {{{{{{rm{V}}}}}}2{{{{{rm{CT}}}}}}}_{{{{{{rm{x}}}}}}}+{{{{{{{rm{NH}}}}}}}_{4}}^{!+}{({{{{{rm{HAc}}}}}})}_{3}+{{{{{{rm{e}}}}}}}^{{{{{{rm{-}}}}}}},({{{{{rm{oxidation}}}}}},, {{{{{rm{PC}}}}}})$$
(5)
The generality in aqueous ammonium ion storage
The generality of this acetate ion enhancement impact on pseudocapacitive capability was additional confirmed in 1T-MoS2-based ammonium-ion battery utilizing a Swagelok-type cell with the layered 1T-MoS2 because the working electrode (Fig. 6a), activated carbon because the counter electrode, and a saturated Ag/AgCl electrode acted because the reference electrode, respectively. 0.5 M NH4Ac electrolyte and 0.25 M (NH4)2SO4 electrolyte had been chosen as two totally different electrolyte methods with the identical NH4+ focus. The GCD profiles in Fig. 6b point out the obvious discrepancy between these two totally different electrolyte methods. The particular discharge capability at 1 A g−1 is 78.0 mAh g−1 in 0.5 M NH4Ac electrolyte, which is way bigger than that in 0.25 M (NH4)2SO4 electrolyte (55.0 mAh g−1). The cyclability of the 1T-MoS2 anode in NH4Ac electrolyte was additionally superior to that in (NH4)2SO4 electrolyte (Supplementary Fig. 27). Moreover, the CV curve space obtained in NH4Ac electrolyte was remarkably bigger than that in (NH4)2SO4 electrolyte at 1 mV s−1 (Fig. 6c). The response course of was additional assessed by CV at totally different sweep charges (Supplementary Fig. 28). In accordance with Eq. (1), the surface-controlled capability was evaluated to be 58.2 mAh g−1 in 0.5 M NH4Ac electrolyte (Fig. 6d), which was clearly increased than the 37.6 mAh g−1 in 0.25 M (NH4)2SO4 electrolyte. All the aforementioned outcomes confirmed the acetate ion enhancement impact can be hanging within the 1T-MoS2-based ammonium-ion battery.
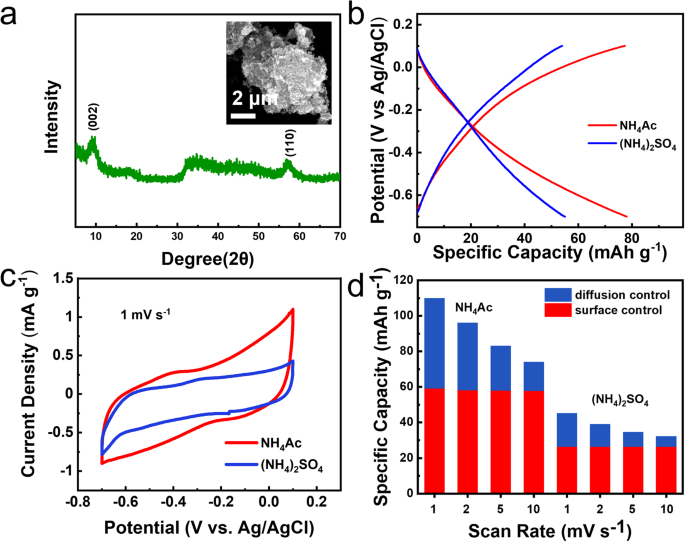
a XRD sample and SEM picture of 1T-MoS2. b GCD profiles of 1T-MoS2 in 0.5 M NH4Ac and 0.25 M (NH4)2SO4 at 1 A g−1. c CV curves achieved in these two electrolytes at 1 mV s−1. d Contributions of 1T-MoS2 in these two electrolytes at assorted scan charges.








