Synthesis and characterization of PVCA
Determine 1a depicts the operational mechanism of quasi-solid-state LMBs produced utilizing the strategically designed PVFH-PVCA electrolyte. Standard liquid electrolyte (LE)-based Li-ion batteries endure from points, corresponding to unstable stable electrolyte interface (SEI) formation and electrolyte decomposition, leading to dendrite development and cathode degradation (Fig. 1a, left)27,28. The PVFH-PVCA electrolyte developed on this research can generate a steady SEI on the Li-metal anode, selling uniform Li deposition. Concurrently, PVCA participates within the formation of the cathode–electrolyte interphase (CEI), suppressing electrolyte decomposition and guaranteeing the structural stability of the LRMO (Fig. 1a, proper). To organize the PVCA ternary copolymer by way of free-radical polymerization, and introduce PVCA into PVFH to type a dense polymer movie, and the corresponding artificial scheme is proven in Supplementary Fig. S1. A tailored multifunctional macromolecule, PVCA, was synthesized by way of a one-step thermally initiated copolymerization utilizing MA, VC, and AN as monomers. Supplementary Fig. S2 reveals the Fourier rework infrared (FTIR) spectrum of the PVCA copolymer. The attribute absorption peaks of the methine (-CH) group stretching vibrations of the AN at 3165, 3126, and 3064 cm−1 disappeared and that of the -CN group appeared at 2236 cm−1. Additional, the attribute stretching vibration peaks of the C=O teams in MA and VC appeared at 1778 and 1341 cm−1. The synthesis of PVCA was confirmed by the absence of the unsaturated C=C moieties within the FTIR spectrum owing to the profitable random copolymerization of AN, MA, and VC. Moreover, the 1H-NMR spectrum of PVCA was analyzed and the peaks within the spectrum had been attributed (Supplementary Fig. 3a). It may be discovered that the distinct peaks appeared across the low discipline (2-3.5 ppm), which might point out the looks of hydrogen atoms related by C-C single bonds. Moreover, the mixing outcomes of the peaks present that the ratio of the variety of substrates concerned within the response in PVAC is MA:VC:AN = 1:3:3. By analyzing the 13C-NMR spectra of the reactants MA, VC, AN, and PVAC (Supplementary Fig. 3b–e), it may be noticed that the peaks of C=C double bonds (100–140 ppm) could be noticed within the reactants MA, VC, and AN. The height depth of C=C double clicking within the product PVAC is considerably diminished, and there’s a clear C-C single bond peak (20-40ppm) within the low discipline, confirming that the substrate did endure polymerization response. As proven in Supplementary Fig. 4a, the molecular weight of PVCA was decided to be 91,100 utilizing Gel Permeation Chromatography (GPC). Thermogravimetric evaluation revealed that the copolymer reveals a single decomposition temperature (Supplementary Fig. S4b).
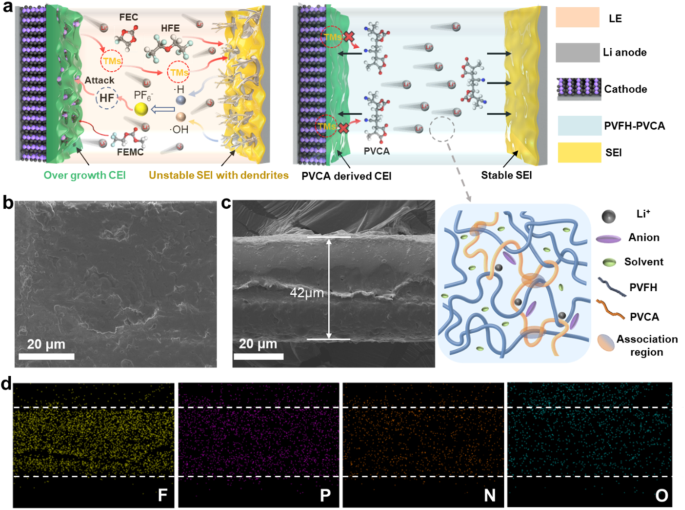
a Schematic illustration of the dendrites fashioned on the Li-metal electrode and degradation processes occurring in cathodes upon utilizing a carbonate-based electrolyte resolution (left) and schematic illustration of the steady stable electrolyte interphase (SEI) fashioned on the Li-metal anode and the inhibited degradation of the cathode interface layer in a Li-metal cell utilizing the PVFH-PVCA polymer electrolyte developed on this research (proper). b and c SEM photographs of the floor b and cross-section c of the PVFH-PVCA polymer electrolyte. d Power-dispersive spectroscopic elemental mapping photographs of F, P, N, and O within the PVFH-PVCA gel polymer electrolyte.
Construction and morphology of PVFH-PVCA blends
To reveal the potential of PVCA as a polymer electrolyte element of high-energy-density LMBs, we chosen PVFH, an electrochemically steady polymer that’s generally utilized in quasi-solid polymer electrolytes, because the polymer matrix. Dynamic mechanical evaluation revealed that PVFH-PVCA membranes with 15 and 20 wt% PVCA have higher mechanical properties (Supplementary Fig. S5) than others. The best conductivity of two.04 × 10−3 S cm−1 at 25 °C was achieved for the pattern with 15 wt% PVCA (Supplementary Fig. S6). Subsequently, a PVCA content material of 15 wt% was used for additional analyses.
Determine 2nd reveals the FT-IR spectra of LE-free PVCA, PVFH, and PVFH-PVCA. The FTIR spectrum of PVFH introduced peaks at 1166 and 1227 cm−1, equivalent to C-F stretching vibrations and -CF2- bending vibrations, respectively. For LE-free PVFH-PVCA, the stretching vibration peak of -CF2- (1227 cm−1) shifted to a better wavenumber whereas that of the -C = O of the carbonate moiety shifted to a decrease wavenumber. Additional, a brand new broad peak appeared at 3415 cm−1, partly as a result of dipole–dipole interplay between the -C = O teams of the PVCA skeleton and the C-F teams of PVFH, which promoted compatibility between the 2 polymers29,30. Then again, the -CN group will increase the electronegativity of the carbon atom of the methine group, favoring robust H···F interactions between the -CH- and -CF2- teams in PVFH, leading to a robust H···F stretching vibration peak31.
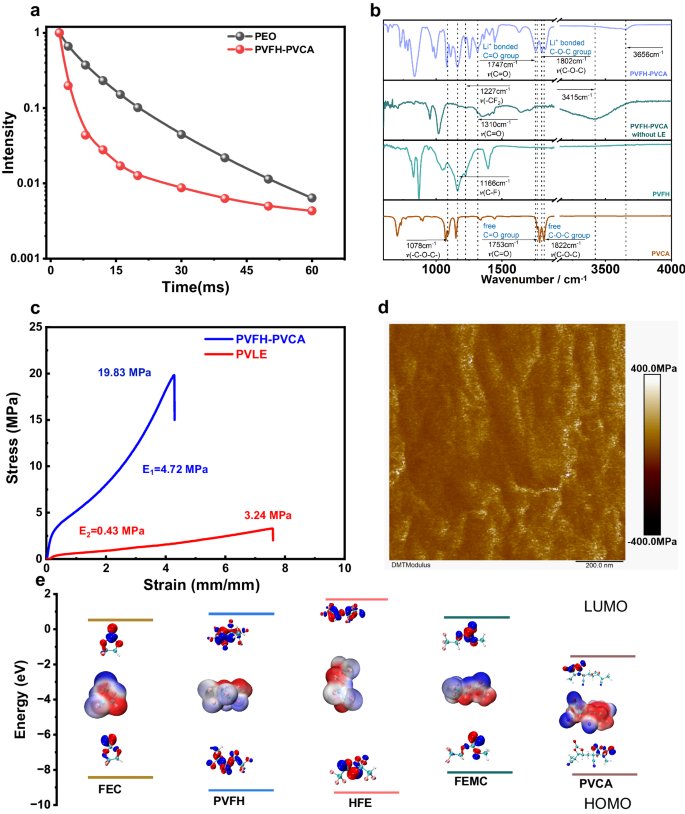
a 1H-NMR T2 leisure curves for PVFH-PVCA and PEO. Adsorption power of PVFH with PVCA ( − 44.461 kJ/mol). b FTIR spectra of PVCA, PVFH, and PVFH-PVCA with out LE and PVFH-PVCA recorded at 25 °C. c Stress–pressure curves of PVFH-PVCA and PVLE at an extension price of 100 mm min−1. d Shear modulus of PVFH-PVCA. e LUMO and HOMO power values of the solvent molecules (FEC, HFE, and FEMC) and polymers (PVFH and PVCA). Insets present the molecular electrostatic potentials and the corresponding visible LUMO and HOMO geometrical buildings.
Interplay between the blended polymers and lithium salt
Upon soaking with the LE, the dry light-yellow PVFH-PVCA membrane step by step became a clear gel at room temperature (Supplementary Fig. S1b). Accordingly, the stretching vibration of H···F shifted to a better wavenumber (Fig. 2b, prime) owing to the formation of hydrogen bonds between the polymer skeleton and fluoroethylene carbonate (FEC) and methyl 2,2,2-trifluoroethyl carbonate (FEMC) within the LE. The high-density H-bonds between the membrane and electrolyte, mixed with the H-bonds between FEC and FEMC and between PVFH and PVCA, trigger the synergistic crosslinking of the 2 polymers at a number of websites, considerably bettering the mechanical energy of the gel electrolyte. Furthermore, the C-O-C and C=O teams coordinate with Li+, as indicated by the shift of their attribute stretching vibrations to decrease wavenumbers32. As well as, the polar -CN purposeful teams within the polymer chain may also work together with Li+, offering a quick Li+-diffusion channel, thereby regulating Li+ distribution throughout lithium deposition/stripping processes, leading to excessive Li+ conductivity33.
PVFH soaked within the LE (PVLE) and Celgard with 60 μL of the LE (CLE) had been chosen as management samples. PVLE has a unfastened, porous floor with a thickness of 47 μm (Supplementary Fig. S7 and S8), whereas the PVFH-PVCA membrane has a notably flatter and extra compact construction attributable to stronger interactions between PVCA and PVFH (see Fig. 1b, c and Supplementary Fig. S9), leading to a excessive adsorption power (Supplementary Fig. S10). Furthermore, each PVCA and LE had been evenly distributed within the PVFH matrix (see Fig. 1d). Moreover, the LE content material within the PVFH-PVCA was ~13.7 wt% (Supplementary Fig. S11); thus, the excessive ionic conductivity of PVFH-PVCA isn’t solely as a result of LE. To additional examine ion coordination within the gel electrolyte, we in contrast the structural traits of PVLE and PVFH-PVCA utilizing Raman spectroscopy. In Li-salt options, cation–anion clusters could be categorized as free anion clusters (FA), loosely sure ion pairs (LIP), and aggregated clusters (AC)34,35,36. As proven in Supplementary Fig. S12, the FA, LIP, and AC contents of PVLE are 40.9, 36.3, and 22.8%, respectively, whereas these of PVFH-PVCA are greater at 57.1, 33.2, and 9.7%, respectively. Thus, PVCA prompts the dissociation of Li salts, resulting in a better variety of cell Li+.
Mechanical and electrochemical properties of PVFH-PVCA
The T2 leisure behaviors of various polymers had been assessed utilizing 1H Hahn echo, acquiring leisure curves of the sign depth as a operate of time on a logarithmic scale (Fig. 2a). To additional validate the entangled construction of PVFH-PVCA, high-molecular-weight polyethylene oxide (PEO) samples with attribute chain entanglement had been chosen as reference samples37. The outcomes revealed a pronounced curvature within the leisure curve of PEO, indicating the presence of a number of and even non-exponential leisure components38. Equally, the comfort curve of PVFH-PVCA indicated fast decay occasions (lower than ~20 ms), equivalent to the movement of entangled chains inside the system, and gradual decay occasions (better than ~30 ms), representing the movement of chain ends and quick chains inside the system. Thus, it may be inferred that PVFH-PVCA not solely shortens the T2 leisure time but additionally transforms the single-exponential decay conduct to a biexponential and even non-exponential one37,39,40.
A freestanding polymer electrolyte membrane with excessive flexibility and mechanical energy is extremely fascinating for bettering compatibility with electrodes and likewise for guaranteeing security throughout operation. Entangled affiliation of polymer chains can successfully improve the mechanical properties of polymer membranes. The PVFH-PVCA membrane exhibited a considerably greater fracture toughness (19.83 MPa) than that of PVLE (3.24 MPa). As well as, the tensile curve indicated that PVFH-PVCA reveals a excessive entanglement phenomenon20. Notably, PVFH-PVCA has a a lot bigger elastic modulus of 4.72 MPa than PVLE (0.43 MPa; Fig. 2c). Moreover, the PVFH-PVCA membrane could possibly be stretched to ~4 occasions its unique size with out being ruptured (Supplementary Fig. S13). This exceptional characteristic could be attributed to the dynamic and reversible fracture and recombination of high-density H-bonds underneath exterior forces, which successfully dissipate power and impart the PVFH-PVCA movie with distinctive toughness. Atomic drive microscopy (AFM) investigations revealed that the PVFH-PVCA membrane has a considerably decrease floor roughness and a a lot bigger Younger’s modulus(188.5 MPa) than PVLE (26.5 MPa, Fig. 2nd and Supplementary Fig. S14). Excessive energy is favorable for inhibiting the expansion of Li dendrites, whereas excessive pressure is important for large-scale fabrication. The mechanism underlying these distinguished adjustments are presumably the presence of sure stiff teams in PVCA, which assemble into a sturdy mechanical part.
Management over the compositions of the SEI and CEI requires that the popular interfacial species ought to have decrease LUMO energies and better HOMO energies than the primary components41,42,43. As proven in Fig. 2e, PVCA has the bottom LUMO power degree and a comparatively excessive HOMO power degree, indicating its potential to preferentially take part within the formation of SEI and CEI. A large electrochemical stability window of a polymer electrolyte is essential for enhancing the power density of LMBs. Linear sweep voltammetry (LSV) was carried out to watch the electrochemical stabilities of PVLE, CLE, and PVFH-PVCA gel electrolytes. In line with the LSV curves in Fig. 3a, the oxidation potentials of PVLE and CLE are related at ~4.7 V vs. Li+/Li, whereas that of PVFH-PVCA is greater (~5.3 V). The elevated oxidation potential signifies the suitability of PVFH-PVCA for high-voltage cathodes. The PVFH-PVCA gel electrolyte exhibited a considerably greater ionic conductivity at 25 °C (2.04 × 10−3 S cm−1) than that of PVLE (0.94 × 10−3 S cm−1; Fig. 3b). Moreover, it introduced a better Li+-transference quantity (0.61) than these of CLE (0.34) and PVLE (0.31) (Fig. 3c and Supplementary Fig. S15), indicating that the migration of anions in PVFH-PVCA was considerably inhibited. On the one hand, a wealthy variety of Li+ coordination (C = O···Li+, -CN···Li+, and C-O···Li+) can enhance the Li+ conductivity, as proven in Supplementary Fig. S16. Then again, the entangled affiliation of the 2 polymers in PVFH-PVCA contributes to the formation of a posh bodily crosslinked community, which may significantly impede anion migration.
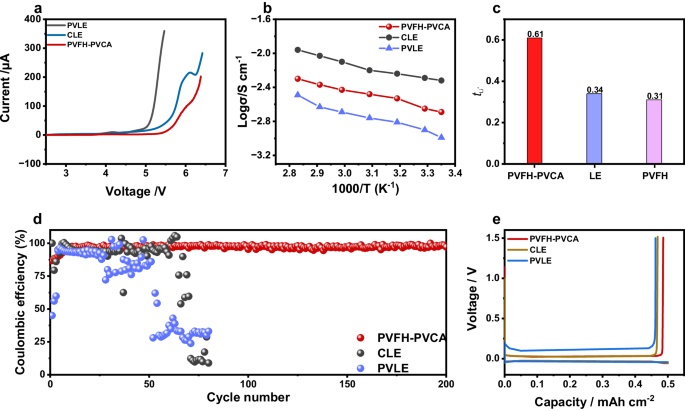
a Electrochemical stability window of PVFH-PVCA, CLE, and PVLE evaluated by linear sweep voltammetry. b Arrhenius plots of PVFH-PVCA and its parts (CLE and PVLE) primarily based on ionic conductivity (σ) versus temperature (T). c Comparability of the Li+-transference numbers of PVFH-PVCA, CLE, and PVLE. d Lithium Coulombic effectivity measured in Li||Cu cells with PVFH-PVCA, CLE, and PVLE. e Voltage profiles of lithium plating/stripping within the Li||Cu cells with PVFH-PVCA, CLE, and PVLE at 0.5 mA cm-2.
The premise of LMB utility is attaining a excessive lithium plating/stripping Coulombic effectivity. The PVFH-PVCA gel electrolyte not solely facilitates the bottom polarization between Li stripping and plating processes (Fig. 3e) but additionally shows a better and extra steady Li Coulombic effectivity over biking (Fig. 3d), indicating an improved interfacial compatibility with the Li anode. This conduct could be attributed to the uniform plating and stripping of the Li metallic in every cycle whereas the superb ion-transport properties can successfully alleviate electrode polarization and stabilize the SEI on the Li anode6.
Li plating and stripping conduct
To validate the speculation, the morphology of electrodeposited Li metallic was characterised by Scanning electron microscopy (SEM). As proven in Fig. 4a, d, the Li| PVLE | Cu cell exhibited a extremely unfastened and porous deposition construction, with a thickness of 49.7 μm, far exceeding the theoretical thickness worth (i.e., 19.4 μm). Porous and fibrous Li metallic was noticed with the CLE (Fig. 4b, e). In distinction, within the cell utilizing PVFH-PVCA electrolyte, a dendrite-free plating construction with compactly aggregated bulk materials was noticed (Fig. 4c). The thickness of the Li layer deposited in PVFH-PVCA electrolyte was solely roughly 21.1μm (Fig. 4f), which was very near the theoretically anticipated worth. These outcomes point out that PVFH-PVCA can deposit Li+ uniformly to type a dense Li metallic layer (Fig. 4g). Such a dense Li deposition with a smaller floor/quantity ratio successfully minimizes the parasitic response between metallic Li and electrolyte44, and thus permits the excessive Coulombic effectivity of Li| PVFH-PVCA | Cu cells (Fig. 3d).

a–f Prime-view and cross-sectional SEM photographs of Li deposits obtained by plating (capability: 4 mAh cm−2) Li on a Cu substrate at 8 mA cm−2 in Li| |Cu cells with a and d, PVLE, b and e, CLE, and c and f, PVFH-PVCA. g Schematic illustration of the Li-deposition conduct in cells with completely different electrolytes.
To analyze the compatibility between the PVFH-PVCA electrolyte and Li metallic, we evaluated symmetric Li||Li cells at 30 °C (Fig. 5a). At a present density of 8 mA cm−2 (capability: 4 mAh cm−2), the voltage curve of the cell with the PVLE confirmed a sustained enhance in polarization, and the cell with CLE failed after ~1100 h due to the formation of an unstable SEI and Li dendrite development. In distinction, the cell with the designed PVFH-PVCA electrolyte could possibly be stably cycled for greater than 4500 h, and the flat voltage plateau throughout plating/stripping remained regular all through the long-term biking interval. The Li||Li cell with the PVFH-PVCA electrolyte could possibly be operated for greater than 1400 h even at a better capability of 5 mAh cm−2 (10 mA cm−2) (Supplementary Fig. S17). Remarkably, no arch form brought on by Li dendrites and useless Li accumulation was noticed on the fringe of the voltage profile. Determine 5b shows the voltage profiles of symmetric Li||Li cells with PVFH-PVCA at numerous present densities within the 1–10 mA cm−2 vary. Owing to the excessive ionic conductivity and glorious interfacial compatibility of the PVFH-PVCA electrolyte, the overpotential of the symmetric Li||Li cell at 10 mA cm−2 was sustained at ~32 mV. In distinction, the PVLE cell was overpolarized at 8 mA cm−2, and the CLE cell failed (Supplementary Fig. S18).
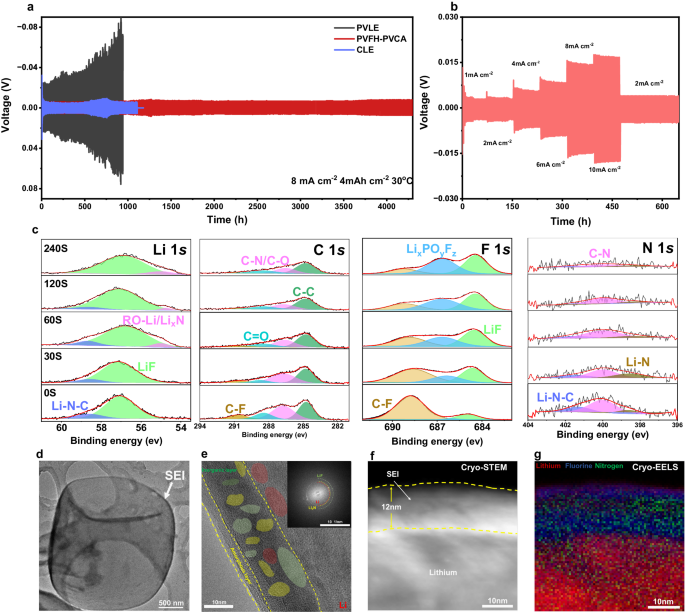
a Galvanostatic voltage profiles of Li||Li symmetric cells with completely different electrolytes at 8.0 mA cm−2 and 4.0 mAh cm−2. b Voltage versus time curves at present densities of 1.0 to 10 mA cm−2 for the PVFH-PVCA cell. c XPS depth profiles of Li 1 s, C 1 s, F 1 s, and N 1 s in Li-metal anodes cycled in Li||Li coin cells with the designed polymer electrolyte. d, e Cryo-TEM photographs of Li deposited within the cell with the PVFH-PVCA at completely different scales. Inset in e reveals the quick Fourier rework sample of the internal SEI: LiF (inexperienced circle), Li (pink circle), and Li3N (yellow circle). f Cryo-STEM picture of Li deposited within the cell with PVFH-PVCA and g the corresponding Cryo-EELS picture.
The morphologies of the Li-metal anodes in cells with completely different electrolytes had been examined to realize insights into SEI formation and the microstructural evolution of the electrodes after 20 cycles at 8 mAcm−2. The floor of the cycled Li anode of the cell with PVFH-PVCA was easy with out black deposits, and the SEM photographs exhibited giant granular, uniform, and compact buildings (Supplementary Fig. S19a and d). In sharp distinction, porous whisker-like Li deposits had been noticed within the SEM photographs of the cycled Li anodes of the cells with CLE and PVLE (Supplementary Fig. S19b and c), and the Li anode surfaces had been lined with black deposits (Supplementary Fig. S19e and f). The less-compact and less-active layer fashioned by “useless” Li, the thick SEI layer, and the porous morphology, along with the depleted Li stock and electrolyte, result in impedance development and untimely cell failure on the anode side45.
A high-resolution X-ray photoelectron spectroscopy (XPS) research of the three electrolytes and Li anode interface additional confirmed the steadiness of the Li interface within the cell with the PVFH-PVCA electrolyte (Fig. 5c and Supplementary Fig. S20, 21). The Li 1 s spectrum of the CLE clearly introduced three parts centered at ~57.0, ~55.8, and ~54.4 eV, that are assigned to LiF, Li2CO3, and LiCOOR, respectively (the place R represents alkyl teams)6,46. As compared, no attribute peak of LiCOOR appeared within the spectrum of PVLE. Within the Li 1 s spectrum of PVFH-PVCA, the Li2CO3 and LiCOOR peaks weren’t detected; as an alternative, two peaks appeared at ~55.0 and 58.9 eV, that are assigned to N-containing species, LixN and Li-N-C13, originating from PVCA. The presence of Li-N-O and LixN was revealed by the N 1 s spectrum, with peaks at ~401.7 and ~398.4 eV, respectively, and a further peak assigned to C-N appeared at ~399.8 eV13. Within the C 1 s spectra of the three electrolytes, peaks of the generally noticed species of C–C (~284.8 eV), C=O (~288.7 eV), and C–O ( ~ 286.1 eV)13,28 had been noticed. Nevertheless, PVFH-PVCA confirmed a better depth of the C=O peak and a further sign of C-F (290.8 eV)47. The presence of C-F (~688.4 eV) was additional confirmed from the F 1 s spectrum48, during which two peaks appeared at ~685 and ~686.4 eV, equivalent to LiF and LixPOyFz43, respectively. The SEI fashioned within the system with PVFH-PVCA had a better content material of F and N (Supplementary Fig. S22). The formation of fluorine-rich and nitrogen-rich interphases contributes to enhanced interfacial kinetics and stability49,50.
We additional investigated the nanostructural options of the SEI utilizing cryo-transmission electron microscopy (Cryo-TEM). Usually, in contrast with the Li dendrites in CLE cells and useless Li in PVLE cells, PVFH-PVCA may induce large deposition of Li (Supplementary Fig. S23); consequently, a steady and uniform SEI was fashioned, as proven in Fig. 5d. When the magnification was elevated to the atomic scale, a dual-layered SEI with an inorganic internal part and an amorphous outer layer within the polymer system was recognized (Fig. 5e). The internal inorganic-rich layer comprised a small quantity of Li3N and a considerable amount of LiF, in step with the XPS outcomes. Cryo-STEM allowed additional visualization of a homogeneous SEI with a thickness of ~12 μm for the PVFH-PVCA cell (Fig. 5f). In distinction, the SEI fashioned within the CLE cell was thicker and inhomogeneous (Supplementary Fig. S24a and c). Cryo-electron power loss spectroscopy (Cryo-EELS) revealed that the SEI was wealthy in N (Fig. 5g and Supplementary Fig. S25) and that F and N had been uniformly distributed within the SEI, which facilitates improved interfacial dynamics and stability, in contrast to within the case of the SEI fashioned by the CLE cell with out N (Supplementary Fig. S24b and S25).
Software of the PVFH-PVCA electrolyte in high-energy-density LMBs
To reveal the applicability of PVFH-PVCA as a potential polymer electrolyte, its electrochemical traits had been studied in LMBs fabricated with numerous cathodes, together with Li1.2Mn0.56Ni0.16Co0.08O2 (LMNCO), LCO, LFP, and NC95. Earlier than evaluating the battery efficiency, electrochemical floating experiments had been carried out on the cells with PVFH-PVCA to precisely decide their precise operational electrochemical home windows (Supplementary Fig. S26)14,51. For the PVFH-PVCA, the measured leakage present was lower than 20 μA as much as a voltage of 4.9 V. Thus, the superb oxidative stability of PVFH-PVCA permits for steady operation (biking and price functionality) with LMNCO cathodes at a excessive voltage of 4.8 V (Fig. 6c, d). The cyclic voltammogram revealed that the LMNCO|PVFH-PVCA|Li battery reveals superior redox response reversibility and kinetics in contrast with these of the techniques primarily based on CLE and PVLE (Supplementary Fig. S27), suggesting that the PVFH-PVCA has glorious interfacial stability with each the LMNCO cathode and Li-metal anode. The biking efficiency revealed that the LMNCO|PVFH-PVCA|Li battery has a capability retention of 84.8% (preliminary capability: 214.1 mA h g−1) after 400 cycles at 2.1–4.6 V and 1 C (Fig. 6a). Evidently, it outperforms different glorious polymer electrolytes developed in earlier works. In clear distinction, the LMNCO|CLE|Li and LMNCO|PVLE|Li batteries exhibited decrease capability retentions of solely 64.0 and 33.9%, respectively, after biking underneath the identical circumstances. As well as, the pristine LMNCO|CLE|Li battery introduced a charge-transfer resistance (Rct) of ~121 Ω, which elevated considerably to ~887 Ω after 300 cycles, whereas the Rct of the LMNCO|PVFH-PVCA|Li battery elevated from ~126 to ~248 Ω after 300 cycles (Supplementary Fig. S28). Determine 6b reveals that the cost/discharge curves of LMNCO|PVFH-PVCA|Li are flat and the voltage polarization stays small after biking. As well as, the capability retentions after 340 cycles at 2.1–4.8 V and 1 C had been as excessive as 78.8% (preliminary capability: 238.4 mA h g−1; Fig. 6c), and the voltage decay was additionally considerably improved (Supplementary Fig. S29). In distinction, the LMNCO|CLE|Li and LMNCO|PVLE|Li batteries had been broken after solely 207 and 100 cycles. The particular capacities of the LMNCO|PVFH-PVCA|Li batteries at 0.1, 0.5, 1.0, 2.0, 4.0, 6.0, and eight.0 C charges are 287.2, 251.1, 215.9, 177.8, 149.1, 126.8, and 109.2 mAh g−1, respectively, a lot greater than these of the LMNCO|CLE|Li battery (Fig. 6d). Extra importantly, the biking and price performances of the LMNCO||Li utilizing the PVFH-PVCA electrolyte had been considerably improved in contrast with these of the batteries with different electrolytes (Supplementary Fig. S30-32).
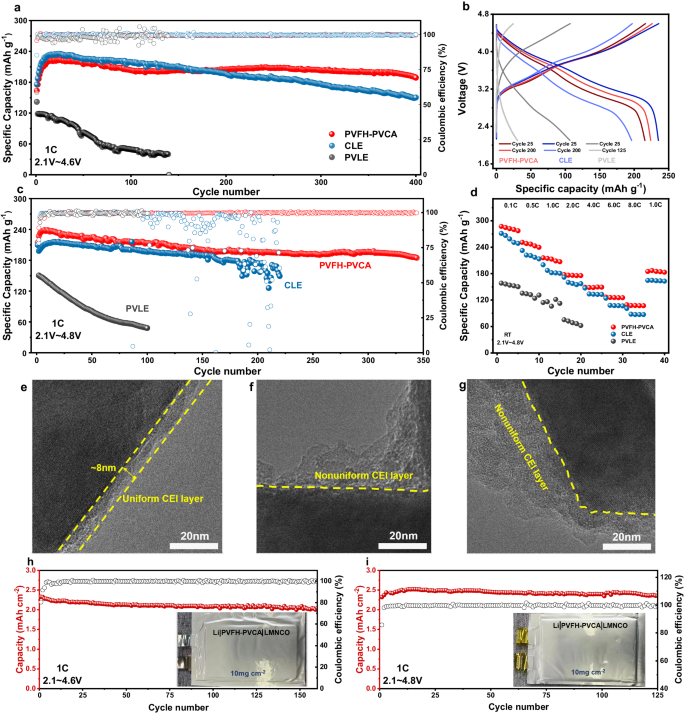
a Biking efficiency of the Li||LMNCO cells with CLE, PVLE, and PVFH-PVCA at 1 C between 2.1 and 4.6 V. b, Cost/discharge curves on the twenty fifth and a hundred and twenty fifth cycles for the Li||LMNCO cells with CLE, PVLE, and PVFH-PVCA at 1 C. c Biking efficiency of the Li||LMNCO cells with CLE, PVLE, and PVFH-PVCA at 1 C between 2.1 and 4.8 V. d, Charge functionality of the Li||LMNCO cells with CLE, PVLE, and PVFH-PVCA from 0.1 to eight.0 C. TEM photographs of the LMNCO particles within the cells utilizing PVFH-PVCA (e), CLE (f), and PVLE (g). h and that i, Lengthy-term biking efficiency of the exemplary Li||LMNCO gentle pack cells using a single layer anode at 2.1–4.6 V (h) and a couple of.1–4.8 V (i).
To grasp how PVFH-PVCA stabilizes the LMNCO cathode, the LMNCO cathode (faraway from the LMNCO||Li button cell after 50 cycles at 1 C and a couple of.1–4.8 V) was sampled and studied in situ. The TEM photographs in Fig. 6e reveal that after biking in PVFH-PVCA, a skinny and uniform CEI layer (~8 nm) was fashioned on the floor of the LMNCO cathode, which facilitated the suppression or delay of intensive intergranular cracking of LRMO particles52 and ensured steady biking of the cell. In distinction, a thicker and non-uniform CEI layer was noticed on the floor of the LMNCO cathode cycled in CLE and PVLE, as confirmed by the TEM photographs in Fig. 6f, g. To additional characterize the chemical states and parts of the CEIs, XPS of the surfaces of the cathodes was carried out after 50 cycles (Supplementary Fig. S33). The peaks of CO32−/O–C = O ( ~ 292.1 eV) and –COOR (287.5 eV) had been clearly noticed within the XPS profiles, indicating undesired electrolyte decomposition on the surfaces of CLE|LMNCO and PVLE|LMNCO. The O 1 s XPS outcomes revealed that the lattice oxygen (529.5 eV) peak of LMNCO on the PVFH-PVCA|LMNCO interface was stronger, indicating {that a} thinner CEI was fashioned on the LMNCO floor, which is in step with the leads to Fig. 6e. Additional, robust C-N alerts had been noticed for the PVFH-PVCA/LMNCO interface, indicative of Li-N-C wealthy inorganic components53.
Though the LMNCO|PVFH-PVCA|Li coin cells exhibit an excellent electrochemical efficiency, excessive loading is fascinating to attain a comparable space capability to that of commercialized cells. Determine 6h reveals a extremely loaded LMNCO|PVFH-PVCA|Li pouch cell cycled at room temperature with an preliminary floor capability of ~2.3 mAh cm−2 (10 mg cm−2) at 1 C and an space capability retention price of 85.2% after 160 cycles. As well as, at greater cut-off voltages, the LMNCO|PVFH-PVCA|Li pouch cell could possibly be operated stably for greater than 120 cycles
To additional confirm the electrochemical efficiency of the PVFH-PVCA electrolyte, LFP|PVFH-PVCA|Li, NC95|PVFH-PVCA|Li, and LCO|PVFH-PVCA|Li full coin cells had been assembled and evaluated (Fig. 7). Excessive-voltage LCO|PVFH-PVCA|Li cells (1 C, 3.0–4.5 V) with a mean discharge voltage of ~4 V exhibited a discharge capability retention of 89.2% after 1500 cycles (Fig. 7a–c). For the LFP|PVFH-PVCA|Li full cell, regular biking stability of 86.4% was obtained after 1940 cycles (0.5 C, 2.6–3.8 V; Fig. 7d–f). As well as, the biking stability of the high-voltage NC95 cathode was additionally improved, and the NC95|PVFH-PVCA|Li cell confirmed 78.7% discharge capability retention (1 C, 2.7–4.3 V) after 200 cycles (Fig. 7g–i). These outcomes verify the universality and compatibility of the PVFH-PVCA electrolyte with numerous electrode supplies.
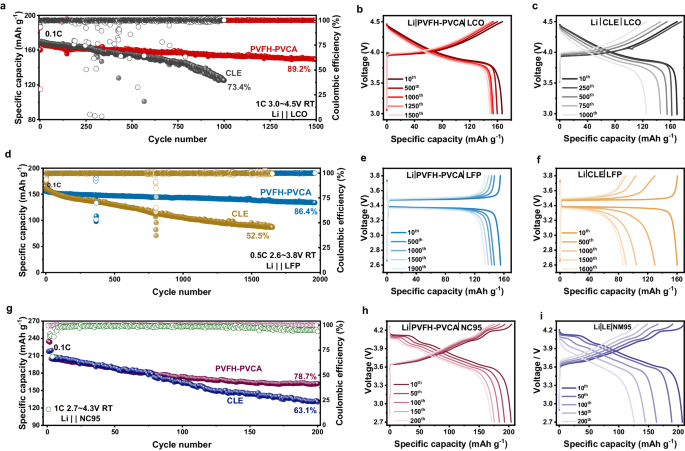
a Comparability of the biking performances of LiCoO2 (LCO)|PVFH-PVCA|Li and LCO|CLE|Li full cells at 1 C. b, c Comparability of the cost/discharge voltage profiles of b, LCO|PVFH-PVCA|Li and c LCO|CLE|Li full cells. d Comparability of the biking efficiency of LiFePO4 (LFP)|PVFH-PVCA|Li and LFP|CLE|Li full cells at 0.5 C. Comparability of the cost/discharge voltage profiles of e, LFP|PVFH-PVCA|Li and f, LFP|CLE|Li full cells. g Comparability of the biking performances of LiNi0.95Co0.05O2 (NC95)|PVFH-PVCA|Li and NC95|CLE|Li full cells at 1 C. Comparability of the cost/discharge voltage profiles of h, NC95 | PVFH-PVCA|Li and that i, NC95|CLE|Li full cells.
In abstract, we efficiently developed an entanglement affiliation polymer electrolyte (PVFH-PVCA) with superior mechanical properties, excessive ionic conductivity, glorious interface compatibility, electrochemical stability, and excessive Li-ion transference quantity by incorporating the PVCA terpolymer. The inherent low oxidation potential and the bottom discount potential of PVCA allow it to preferentially oxidize and type a skinny, uniform, and strong CEI on the LMNCO cathode floor; this successfully suppresses facet reactions with the electrolyte. Moreover, the preferential participation of PVCA in SEI formation on the Li anode results in a dual-layered SEI enriched in N and F, which differs considerably from the standard SEI construction. Thus, it inhibits the formation of “useless” Li and promotes dendrite-free Li deposition, thereby enabling symmetrical Li//Li batteries to attain a exceptional biking stability for as much as 4500 h at a excessive present density of 8 mA cm−2. Moreover, the Li| PVFH-PVCA|LMNCO batteries exhibit glorious biking stability for greater than 340 cycles at a cut-off voltage of 4.8 V. Additional, high-loading Li||LMNCO pouch batteries additionally exhibit excellent biking and a excessive floor capability. The PVFH-PVCA hybrid design has important deserves when it comes to compatibility and could be prolonged to different standard cathode supplies to comprehend glorious cycle stability. This research offers a brand new avenue for high-energy-density Li–LRMO batteries.








