Idea of coupling RMORs with CRR
Our aqueous Zn-CRR/RMOR battery makes use of a Fe-N-C catalyst cathode and a Zn plate anode, separated by a bipolar membrane. A 0.5 M CO2-saturated KHCO3 resolution and a 1 M KOH + 0.2 M Zn(CH3COO)2 resolution have been employed as catholyte and anolyte, respectively. As illustrated in Fig. 1a, throughout discharge, the conversion of CO2 (e.g. from pretreated flue fuel) to CO happens within the cathode compartment whereas the Zn plate is dissolved into the anolyte, affording a voltage of 0.5 V at 0.71 mA cm−2. Throughout cost, the lowering molecules are oxidized to generate N2 fuel, acetone or sulfate, and the Zn2+ is plated again to the Zn anode, finishing replenishment of the battery.
In distinction to the conventionally configured Zn-CO2 battery that depends on high-potential and kinetically sluggish OER on cost, by utilizing tamed RMOR, our Zn-CRR/RMOR battery maintains the chemical integrity of the FeN4 energetic websites, due to this fact exhibiting excellent stability with a low charging voltage (Fig. 1b). The uneven redox reactions allow synergistic CO2 conversion, power storage, and wastewater therapy that may be coordinated each day in follow, right here taking a cement-making manufacturing facility for instance. Within the day, when carbon emission and energy calls for are intensive, the Zn-CRR/RMOR system undergoes discharge thus outputting power and changing CO2 to chemical substances, whereas within the night time, the system recharges to oxidize hydrazine and to replenish the zinc anode. Based on our preliminary techno-economic evaluation, this know-how reveals apparent financial benefits in comparison with that of the classical LiFePO4 battery, the Zn-CO2 battery, and the electrochemical CRR system (see Supplementary Fig. 1 and Observe 1 for extra particulars).
To realize this aim, we recognized and optimized an atomically dispersed Fe-N-C catalyst for catalyzing the discount of CO2 to CO, and the oxidation of lowering molecules. The detailed synthesis and bodily characterizations are mentioned in Supplementary Figs. 2–4, Desk 1, and Observe 2. Gaseous and liquid merchandise have been quantitatively analyzed by utilizing on-line fuel chromatography (GC) and 1H nuclear magnetic resonance (NMR), respectively. For the standard CRR machine (three-electrode cell), the results of product evaluation demonstrates that solely gaseous merchandise (H2 and CO) will be detected with none liquid product (Supplementary Fig. 5). The obtained Fe-N-C catalyst demonstrates exceptional selectivity for CO era at low over-potentials with FECO over 90% from −0.3 to −0.6 V vs. the reversible hydrogen electrode (RHE, used all through the article), with peak FECO of 98% at −0.4 V vs. RHE (Supplementary Fig. 5b). For developing the aqueous Zn-CRR/RMOR batteries, we word that growing the catalyst loading mass reveals little correlation with FECO, albeit resulting in a slight improve in voltages (Supplementary Fig. 6 and Observe 3). For a complete analysis, the loading mass is established to be 1 mg cm−2. The assembled zinc battery reveals a discharging voltage of 0.5 V and a FECO as excessive as 98% at present densities starting from 0.71 to 12.86 mA cm−2, confirming excessive CRR performances.
Basic catalytic conduct with the lowering molecules
We first examined the influence of the lowering molecules on the Fe-N-C catalyzed CRR and the oxidation reactions in three-electrode cells utilizing the linear sweep voltammetry (LSV). The presence of lowering molecules reveals slight influence on the CRR present response with saturated CO2 (Fig. 2a). For oxidation, we denote the oxidation of Hz, ACTO, and TUDO as HzOR, ACTOR, and TUDOR, respectively. For a present density of 10 mA cm−2, HzOR reveals an oxidation potential of solely 0.38 V, and ACTOR and TUDOR present 1.18 V and 1.25 V, respectively, all of that are decrease than 1.57 V for OER (Fig. 2b). Observe that the distinction in exercise of those oxidation reactions will be defined by the cost switch functionality as assessed by the electrochemical impedance spectroscopy (EIS)29,30. The charge-transfer resistance (Rct) of HzOR is just about 1/3, 1/4, and 1/8 of that in ACTOR, TUDOR, and OER (Fig. 2c). A rise within the focus of lowering molecules can decrease the oxidation potential and improve the present response, significantly for HzOR (Supplementary Fig. 7).
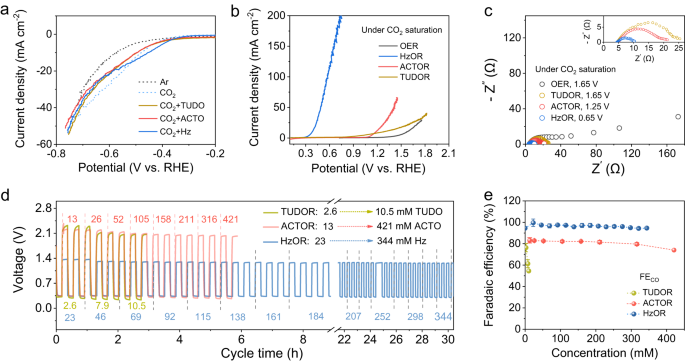
a LSV curves of Fe-N-C catalyzed CRR with out and with the presence of lowering molecules (Ar saturated resolution is included for reference and CO2 is saturated in all different circumstances). b LSV curves of Fe-N-C catalyzed oxidation reactions with totally different lowering molecules vs. OER. c EIS plots obtained with totally different oxidation processes at respectively famous potentials. Analysis of the Zn-CRR/RMOR batteries voltages (d) and FECO (e) in response to assorted concentrations of the lowering molecules. The numbers famous in d signify concentrations of TUDO (in yellow), ACTO (in pink), and Hz (in blue); the error bars in e are the usual deviations from a number of measurements.
For developing the Zn-CRR/RMOR batteries, the influence of lowering molecule concentrations on the voltages and FECO was additionally investigated (Fig. second, e), whereby the gaseous merchandise have been periodically quantitated by way of on-line GC. Whereas the FECO decreases from 77% to 54% as TUDO focus will increase from 2.6 to 10.4 mM, the voltages and FECO will be well-maintained inside a variety of concentrations of ACTO (13–334 mM) and Hz (23–344 mM). Notably, excessive FECO of 97% and really steady voltages will be retained for HzOR even when the Hz focus will increase by 15-folds (Fig. second, e). Curiously, as Hz focus rises to 459 and 688 mM, the charging voltage will be additional lowered however with a lower in FECO to 80% and a lower in output voltages to 0.25 V (Supplementary Fig. 8).
Efficiency of the Zn-CRR/RMOR batteries
Our Fe-N-C catalyst displays wonderful long-term sturdiness in a unidirectional CRR cell (as much as 237 h with FECO over 95%) or OER cell (Supplementary Fig. 9). Nevertheless, a full Zn-CO2 battery involving OER reveals a really brief biking life and speedy decline in FECO (Fig. 3a, b). In nice distinction, by changing OER with RMORs, the Zn-CRR/RMOR battery reveals superior rechargeability and excessive FECO of 92% over 1000 cycles, as mentioned under (Fig. 3a, b).
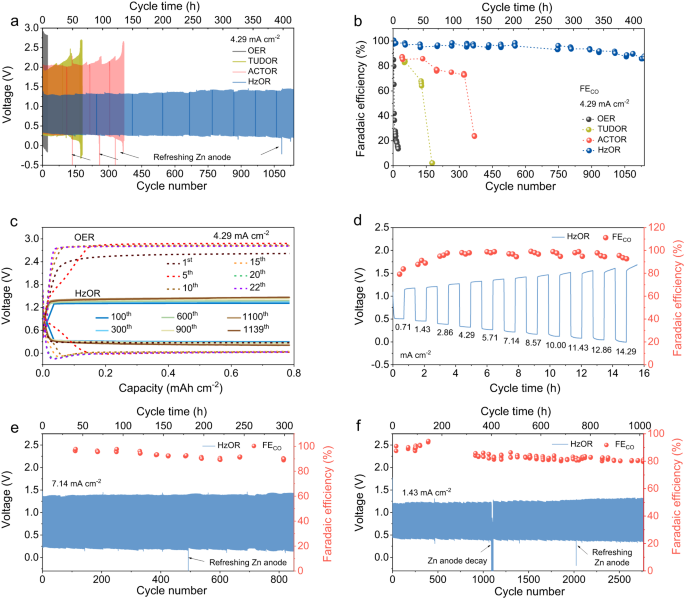
The battery voltage evolution (a) and the corresponding FECO (b) of the Zn-CRR/RMOR batteries with assorted RMORs at 4.29 mA cm−2 (with OER because the reference). The preliminary concentrations of TUDO, ACTO, and Hz in a and b are 7.9, 79, and 92 mM, respectively, and their every day addition quantity are 5.2, 26, and 46 mM. c Galvanostatic discharge/cost voltage profiles exhibiting a a lot decrease voltage polarization in Zn-CRR/HzOR batteries than within the Zn-CO2 battery involving OER. d The speed functionality measurements exhibiting the voltages of the Zn-CRR/HzOR battery with assorted present densities (69 mM Hz). Lengthy-term biking efficiency of the Zn-CRR/HzOR battery demonstrating continued excessive selectivity for CO and voltage stability at 7.14 mA cm−2 (e) and 1.43 mA cm−2 (f). The preliminary focus of Hz in e and f is 92 mM, and the every day addition quantities are 23 and 46 mM for e and f, respectively. The FECO was periodically measured for all methods to make sure a correct understanding of the temporal evolution of CRR; the arrows point out when the Zn anode and anolyte are refreshed, which within the case of HzOR happens each ten, seven, 5 days for measurements at 1.43, 4.29, and seven.14 mA cm−2, respectively. For all batteries, every cycle consists of 11 min cost and 11 min discharge.
At 4.29 mA cm−2, HzOR reveals a a lot decrease charging voltage (1.35 V) than OER (2.45 V), and so are TUDOR (2.0 V) in addition to ACTOR (2.1 V) (Fig. 3a). The extraordinarily steady voltage evolution for the batteries utilizing RMORs is illustrated in Fig. 3a, c and Supplementary Fig. 10. Additional, with OER on recharging, the FECO rapidly decreased from 98% to 80% and retained solely 14% after 22 cycles (Fig. 3b). In sharp distinction, the periodically measured FECO for the Zn-CRR/HzOR battery maintained about 97% and 92% FECO at 100 and 1000 cycles (Fig. 3b). Observe that the biking life obtained for the Zn-CRR/RMOR batteries follows the order of HzOR (1140 cycles, 418.0 h) >ACTOR (369 cycles, 135.3 h) >TUDOR (180 cycles, 65.8 h). As well as, the presence of Zn2+ in catholyte has little impact on the electrochemical conduct of our Zn-CRR/HzOR battery, thus eliminating the priority on potential Zn2+ crossover throughout cycles, as mentioned in Supplementary Fig. 11 and Observe 4. Additional, the Zn-CRR/HzOR battery presents superior charge functionality together with a powerful selectivity towards CO era (Fig. 3d). Excessive FECO of ~96% in a variety of present densities from 2.86 to 14.29 mA cm−2 is achieved. Additionally, the charging voltage rises from only one.15 to 1.70 V when present density will increase by 20-folds, demonstrating wonderful response kinetics for CRR and HzOR (Fig. 3d). Determine 3e reveals {that a} fascinating FECO of 90% over 850 cycles will be achieved at a excessive present density of seven.14 mA cm−2. Notably, the battery reveals an excellent biking lifetime of 2768 cycles (1015.8 h) at a average present density of 1.43 mA cm−2 (Fig. 3f), with excessive FECO retention (90% of the preliminary FECO) and regular voltages (cost: 1.35 V; discharge: 0.38 V; Supplementary Fig. 12). These matrixes are far past the attain of any reported Zn-CO2 batteries21,22,23,24,25,26,27,29,30,31,32,33, with extra particulars summarized in Supplementary Desk 2. All of the discharge and cost merchandise are decided utilizing on-line GC and 1H NMR measurements, as summarized in Supplementary Desk 3, Figs. 13–15, and Notes 5, 6. As well as, the Faradaic effectivity of H2 over biking at varied present densities are additionally supplied in Supplementary Fig. 16.
Such wonderful electrochemical efficiency will be attributed to the low potential of RMOR that stops the catalyst from degradating at excessive potential and guarantee structural stability and environment friendly CRR. Following the identical technique, we additionally show the success of selling one other kind of CRR by coupling with RMORs, as showcased by the CO2 discount to formic acid with a a lot increased FE utilizing a Bi-based catalyst (Supplementary Fig. 17). This confirms the generalizability of our configuration to quite a lot of CRRs the place RMORs suppresses degradation of the catalysts. It’s identified that zinc anode is stricken by the dendrite development and hydrogen evolution (Supplementary Fig. 18). Herein as a substitute of huge dendrites, we noticed the formation of mossy fibrous deposits with a small measurement within the Zn||Zn symmetric battery (Supplementary Fig. 19c, d). To additional enhance the zinc anode and essentially alter its plating conduct, we formulated a functionalized electrolyte (3 M KOH + 1.4 M KF + 0.75 M K2CO3 + 0.032 M ZnO, denoted as 3 M KOH + KFCZnO) that helps a way more uniform and flatter Zn stripping/platting within the Zn||Zn symmetric battery and Zn-CRR/HzOR battery (Supplementary Figs. 19e, f, and 21d–f). As a result of suppressed hydrogen evolution, much less lifeless zinc, and decrease focus polarization, enhanced charge capabilities with steady biking efficiency have been obtained. Particularly, the discharged voltage was promoted from 0.32 V to 0.44 V at 4.29 mA cm−2 (see Supplementary Figs. 18–21 and Notes 7–10 for extra particulars).
Mechanistic insights on enhanced CRR exercise
To disclose the mechanism of lowering molecules impact on CRR, density practical idea (DFT) calculations have been carried out to visualise the CRR course of on FeN4 exercise facilities (the FeN4 configuration is confirmed under). The damaging adsorption power of ACTO (−0.72 eV) and Hz (−0.74 eV) signifies that their adsorption on the catalyst floor is energetically favorable. We first present that the much less distorted FeN4 websites upon adsorbing *COOH and *CO with the presence of Hz or ACTO account for the excessive CRR selectivity (mentioned in Fig. 4a, Supplementary Fig. 22, and Observe 11). Following the adsorption and activation of CO2 molecules, the clean FeN4 websites apparently present a bigger distortion, which leads to a stronger interplay between Fe and *CO, making it troublesome to launch CO. In distinction, with the presence of Hz or ACTO, there’s much less distortion of the FeN4 web site indicating weaker interplay with the *CO that drives its facile desorption. That is owed to the electron-donating characteristic of the lowering molecules added, which implies that the technique employed right here synergistically advantages the low-potential oxidation and high-activity CRR. That is essential for the success of our idea of coupling RMOR with CRR in a single bidirectional battery.
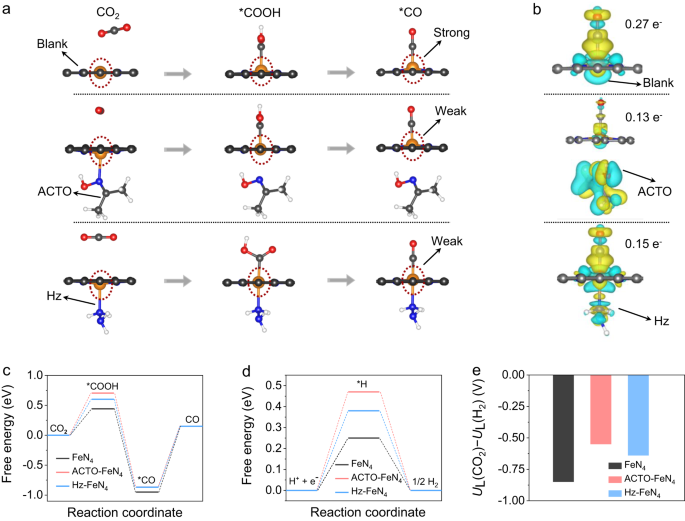
a The calculated FeN4 web site configurations with lowering molecules adsorbed throughout the CRR course of (orange, blue, grey, pink, and pink spheres stand for Fe, N, C, O, and H atoms). b The calculated cost density distribution of CO* adsorbed on FeN4 websites (yellow and cyan areas signify accumulation and depletion of cost). c–e Calculated free power diagrams for the CRR (c) and HER (d) processes on the FeN4 websites with or with out the lowering molecules, and the corresponding UL(CO2) − UL(H2) values (e), an indicator for CRR selectivity.
The discovering is additional supported by the differential cost density analyses of the *CO adsorbed on the FeN4 websites whereby the cost switch (between CO and FeN4) is much less in Hz-FeN4 (0.15 e−) and ACTO-FeN4 (0.13 e−) than in clean FeN4 (0.27 e−) (Fig. 4b). A much less cost density switch means a weaker binding power for CO and therefore a decrease power barrier for its desorption22. We word that ACTO loses it binding to the Fe atom as soon as COOH* and CO* are shaped, whereas Hz maintains the shut binding with Fe all through the CRR course of, thereby exhibiting the least distortion of FeN4 websites amongst all methods studied.
In keeping with earlier reviews, we additionally discovered that CRR on the FeN4 websites is ruled by the desorption of *CO as a result of robust binding between *CO and Fe (Fig. 4c)25,26,31. With the presence of Hz or ACTO, *CO desorption reveals a barely decrease power barrier than that within the clean FeN4 (1.02 eV vs. 1.10 eV), indicating the lowering molecules don’t incur additional power penalty and will barely promote the intrinsic exercise of CO2-to-CO discount. Moreover, HER as a dominant competing response upon CRR, was investigated. A better power barrier for the formation of H* is noticed on the FeN4 web site with the presence of Hz and ACTO (Fig. 4d), indicating that Hz and ACTO have an inhibiting impact on H2 era. We additional quantified the limiting potential distinction between CRR and HER, i.e. the UL(CO2)–UL(H2), an indicator for CRR selectivity (Fig. 4e)21,26. In comparison with the clean FeN4 (–0.88 eV), extra optimistic values for the Hz-FeN4 (–0.7 eV) and ACTO-FeN4 (–0.6 eV) websites counsel the upper selectivity for CO formation21,26. As for TUDO, our proton-releasing experiments counsel that the H+ launched from TUDO results in a decrease FECO in comparison with that on HzOR-FeN4 and ACTOR-FeN4 websites (Supplementary Fig. 23 and Observe 12).
Mechanistic insights on the long-term stability
We then focus on the mechanism of the considerably promoted long-term stability of the batteries. First, in nice distinction to the virtually an identical voltage profiles (first 4 cycles) for the Zn-CRR/RMOR batteries, the one with OER reveals a sudden improve in charging voltage on the third cycle (2.4–2.8 V) (Fig. 5a, Supplementary Fig. 24). That is clearly proven by an extra oxidation peak within the dQ/dV curve (Fig. 5b). We attribute such phenomenon to the degradation of catalyst, that’s, electrochemical Fe demetallation occurring throughout OER, which has been present in ORR/OER systems34,35,36,37. Particularly, the reactive oxygen species OH* produced throughout OER first adsorbs on FeN4 websites, and the OH* weakens Fe–N bonding and triggers the leaching of Fe34,35. To confirm the soundness of FeN4 websites in opposition to demetallation, we assembled batteries that recharge by alternating OER and HzOR (Supplementary Fig. 25). In all circumstances when OER was launched, a pointy decline in FECO occurred; and critically, even when HzOR was launched again on cost, the discharging voltage and FECO remained extraordinarily low, suggesting everlasting injury to the catalyst by OER. We thus unambiguously present this results in its irreversible sharp decline within the CRR exercise over biking of a Zn-CO2 battery.
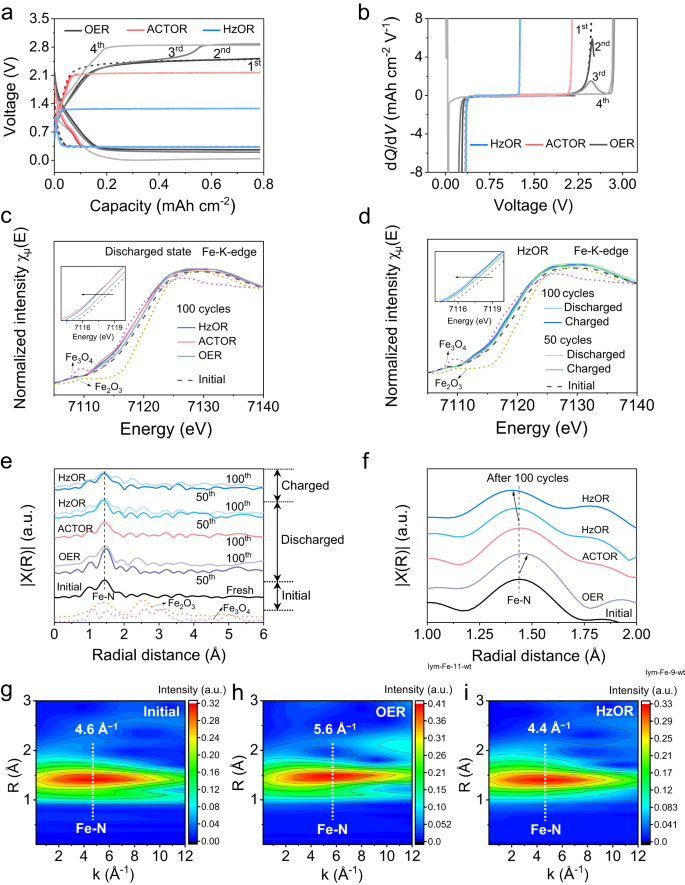
Preliminary 4 galvanostatic discharge/cost profiles (a) exhibiting a sudden improve in charging voltage for the OER-based battery, and their corresponding dQ/dV profiles (b) throughout charging. c Fe Okay-edge XANES spectra of Fe-N-C indicating a extra optimistic valence of Fe in OER-based cathode at discharged state. d Fe Okay-edge XANES spectra of HzOR-based cathodes at charged and discharged states. The insets of (c) and (d) are the enlarged Fe Okay-edge XANES spectra. e, f Fourier transformation (FT)-EXAFS spectra of cathodes after biking, displaying Fe-N coordination with magnification in f. g–i Fe wavelet rework (WT)-EXAFS spectra of Fe for the preliminary, OER- and HzOR-based cathodes after 50 cycles; the colour bar represents the depth.
We carried out Fe Okay-edge X-ray absorption spectroscopy (XAS) research to look at the native chemical and structural evolution of the Fe-N-C catalyst over long-term biking. The X-ray absorption near-edge construction (XANES) spectrum for the preliminary cathode locates at energies between these of Fe2O3 and Fe3O4, indicating the common valence of Fe in Fe-N-C is between +2 and +3. Finding at a extra optimistic power signifies a extra oxidized state for the Fe37,38. After 100 cycles, the near-edge of the OER-based cathode locates at a better power than HzOR- and ACTOR-based cathodes, suggesting increased oxidation state of Fe (Fig. 5c, Supplementary Fig. 26 and Observe 13). We additional observe a pattern of shifting to decrease power alongside biking for the HzOR-based cathode, showcased by the spectra after 50 and 100 cycles (Fig. 5d). This means that Fe is additional proof against being oxidized over biking, which will be ascribed to the lowering atmosphere launched by CRR38. The evolution of Fe valence is expounded to the structural adjustments, as additional mentioned under. Furthermore, the Okay-edge spectra for HzOR-based cathodes at charged states virtually overlap with these at discharged states, suggesting wonderful catalyst stability that’s proof against each discount and oxidation (Fig. 5d).
The prolonged X-ray absorption high-quality construction (EXAFS) fittings additional reveal the native structural configuration of Fe within the catalyst. The k- and R-space spectra of the preliminary catalyst are well-fitted with atomic dispersion of Fe coordinated with 4 N atoms within the first shell, indicating a predominant FeN4 mode in Fe-N-C (Supplementary Fig. 27 and Desk 4)21,26. Over biking, the OER-based cathode reveals a better Fe-N bonding size (1.48 Å) whereas the ACTOR- and HzOR-based cathodes present comparable and even shorter Fe-N bonding lengths than the pristine (1.44 Å, 1.42 Å; Fig. 5e, f, fitted spectra in Supplementary Fig. 27). That is additional confirmed by the wavelet rework (WT)-EXAFS analyses (Fig. 5g–i, Supplementary Fig. 28). Clearly, the k-space depth most for the ACTOR-cathode (4.6 Å−1) and HzOR-cathode (4.4 Å−1) is near that for the preliminary cathode (4.6 Å−1), in nice distinction to the one for OER-based cathode (5.6 Å−1; Fig. 5g–i, Supplementary Fig. 28). Such distinction could possibly be associated to the refined variations within the native coordination environments of Fe atoms in these cathodes28,36,39,40, as additional mentioned in Supplementary Observe 13. The truth is, the Fe-N-C catalyst comprises two forms of FeN4 websites, the pyridinic N-coordinated web site (denoted as S1) and the pyrrolic N-coordinated web site (denoted as S2)34,38,41. For the OER-based cathode, the interplay of Fe (of S1) with *OH radicals causes its Fe demetallation upon biking (Fig. 5a, b)34,35, leaving majorly S2-Fe. Such coordination possesses a better Fe oxidation state38, explaining the XANES outcomes (Fig. 5c), and the remaining low-activity S2 websites present poor selectivity for CRR.
We additional experimentally verify the Fe demetallation by quantifying the presence of iron within the electrolytes from cells performing remoted OER, RMORs or coupled CRR-RMOR (Supplementary Tables 5–7, and Notes 14–16). Notably, the orange-colored electrolyte after OER in distinction to the colorless electrolyte after HzOR supplies direct visible proof (Supplementary Fig. 29). Observe that the scanning electron microscopy (SEM) photographs of the cathodes after CRR and varied battery operations present inherited nanoparticle morphology, implying that the carbon framework of Fe-N-C is nicely preserved (Supplementary Figs. 30 and 31).
One dangling query is why the Zn-CRR/HzOR battery nonetheless reveals excessive efficiency regardless of a slight diploma of Fe demetallation (Supplementary Tables 5–7). We right here suggest two assumptions: a) the Fe demetallation might happen on the S2 websites proudly owning to the electron-donating characteristic of Hz molecules and the low charging voltage, and thus the S1 websites stay majorly energetic; and b) even when Fe demetallizes from S1 websites, electrochemical transformation of the S2 websites to S1 websites might happen. Each assumptions are potential as indicated by a shorter Fe-N distance (1.42 Å) and fewer oxidized state of Fe. Additional examine into these assumptions is required sooner or later. Taken collectively, extremely energetic Fe websites in Fe-N-C account for the long-term operation of the Zn-CRR/HzOR battery.
Scalability demonstration
There was an absence of demonstration for scalability within the subject of Zn-CO2 batteries. Provided that our Zn-CRR/HzOR battery dramatically outperforms all reported Zn-CO2 batteries by way of the biking life, charging voltage, and FE of the aimed product (Fig. 6a, b, and Supplementary Desk 2)21,22,23,24,25,26,27,29,30,31,32,42,43,44,45,46,47,48,49, we now show the scalability of our battery that {couples} CRR and HzOR.
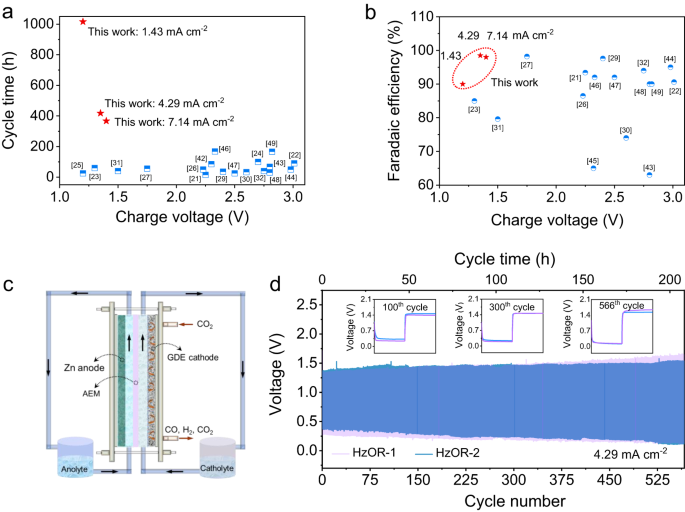
The electrochemical efficiency of the Zn-CRR/HzOR batteries that drastically outperforms the reported Zn-CO2 batteries, relating to the charging voltage as a perform of cycle time (a) and the FE of CO or formic acid era (b). c Schematic of the circulation cell. d The long-term stability of two practically an identical circulation cells based mostly on HzOR (HzOR-1 and HzOR-2), indicating excessive reproducibility within the vary of used circulation charges: the circulation charge of catholyte have been 13.4 and 26.8 rpm for HzOR-1 and HzOR-2, respectively, and the anolyte circulation charge remained at 20 rpm.
For such, a scaled circulation cell configured with a fuel diffusion electrode (GDE) and an anion change membrane (AEM) was assembled, as illustrated in Fig. 6c. The dynamic equilibrium between CO2 fuel and catholyte is important for the sturdiness of GDE. The cells utilizing optimized catholyte circulation charges (13.4 and 26.8 rpm) are denoted as HzOR-1, and HzOR-2. Each cells confirmed a wonderful rechargeability with a small discharging–recharging voltage polarization of ~1.0 V over a steady operation for 566 cycles (207.7 h, Fig. 6d). The unprecedented efficiency proven right here signifies the practicability and scalability of our Zn-CRR/HzOR battery.
Choice standards of lowering molecules for future design
A prerequisite for the molecules is the strongly lowering nature, which is evidenced by the speedy fading of the investigated Zn-CRR batteries which might be based mostly on the oxidation of non-reducing methanol, ammonia, and urea (Supplementary Fig. 32 and Observe 17). Along with the lowering molecules of Hz, ACTO, and TUDO mentioned above, one other two lowering molecules, butanone oxime (BTO) and acetaldoxime (AAO) present enhanced biking efficiency within the Zn-CRR/RMOR batteries, illustrating the generality of lowering molecules results (Supplementary Fig. 33).
The second consideration is that the oxidation merchandise of lowering molecules ought to preferentially be simply separated and picked up from the system in real-time. Our Zn-CRR/HzOR battery reveals an excellent biking life with out the necessity for periodic resolution purification owing to the gaseous product N2 that endows integrity of the system (detailed dialogue in Supplementary Figs. 13, 14, and Observe 5). A much less favorable case is the formation of carbonaceous organics similar to acetone and urea, which may poison the energetic websites and trigger the battery failure ought to no periodic regeneration be put in (particulars in Supplementary Figs. 13, 15, 34, and Notes 5, 6, 18).
The opposite requirement is that the oxidation potential of lowering molecules must be as little as potential for top power effectivity, which is essential for this new know-how to realize synergistic CO2 utilization and power storage in an energy-economical method.








