Construction characterizations and electrochemical behaviors of CoSx
The CoSx (x = 8/9, 1.097, and a pair of) samples had been synthesized by sulfidation of Co(OH)2 nanosheet arrays electrodeposited on items of carbon papers (CP) (Supplementary Fig. 1). Their X-ray diffraction (XRD) patterns in Supplementary Fig. 2 comprise diffraction peaks that match these of ordinary CoS2, CoS1.097, and Co9S8 apart from peaks at about 26°, 43° and 55°(2θ), which correspond to the CP substrate7,14,48. The morphologies of the as-prepared CoSx had been studied utilizing scanning electron microscopy (SEM). Supplementary Fig. 3a–d present that CoS2, CoS1.097, and Co9S8 keep the same nanosheet construction to electrodeposited Co(OH)2. Supplementary Fig. 4 additionally confirms that the electrochemical floor lively space (ECSA) of the three cathodes is analogous, ruling out their affect on the next electrochemical take a look at. The high-resolution transmission electron microscopy (HRTEM) pictures in Supplementary Fig. 5a–c present three units of lattice fringes, with interplanar spacings of 0.28 nm, 0.29 nm, and 0.30 nm, which might be assigned to (200), (204), and (311) planes of CoS2, CoS1.097, and Co9S8, respectively. Component mappings in Supplementary Fig. 6a–c affirm the uniform distribution of Co and S. Their electrochemical behaviors exhibit notable variations in Li-CO2 batteries, as proven in Fig. 1c–e and Supplementary Fig. 7. Batteries with Co9S8 cathodes present a rise in overpotential, whereas the discharge and cost curves for the CoS1.097 cathodes solely have a slight change in 10 cycles. Curiously, the cost plateau of CoS2 for 10 cycles is way decrease than the pristine one, and the overpotential is decreased from 0.89 V to 0.52 V. These variations immediate our investigation into the CO2 response and actual lively construction inside Li-CO2 batteries.
The structural evolution characterizations
To research structural evolutions that have an effect on the electrochemical behaviors, we carried out X-ray photoelectron spectroscopy (XPS) and X-ray absorption spectroscopy (XAS) analyses on the cathodes at totally different cycles. The XPS ends in Supplementary Figs. 8 and 9 point out that S in CoS2 has lower than the total coordination, which maintains its construction throughout biking in Li-CO2 batteries. In distinction, Co-S bindings lower considerably in each CoS1.097 and Co9S8, suggesting extreme structural adjustments throughout biking. To additional affirm the precise construction after biking, we carried out XAS at Co Ok-edge to check the evolution of Co9S8, CoS1.097, and CoS2 earlier than and after 10 cycles (labeled as CoSx-10) in Fig. 2a–c. X-ray absorption near-edge construction (XANES) in Fig. 2a, b present that the absorption edges of Co9S8 and CoS1.097 shift to greater power and overlap with that of CoO after biking, indicating sulfide oxidation. Fourier remodeled prolonged X-ray absorption effective constructions (FT-EXAFS) in Fig. second, e present that the primary coordination shell of Co9S8-10 and CoS1.097-10 is way shorter than that of the pristine samples and are near that of CoO, confirming most S atoms being substituted by O atoms. In distinction, for CoS2, the absorption fringe of CoS2-10 is situated between that of pristine CoS2 and CoO, and the primary coordination shell of Co is contracted however nonetheless longer than that of Co-O (Fig. 2c, f). We examine the FT-EXAFS of CoS2-10 with FEFF49-calculated Co-O path and Co-S path, displaying that the oscillation within the first coordination shell of CoS2 cannot be solely assigned to Co-O scattering or Co-S scattering (Supplementary Fig. 10 a–c). The factor of scattering atoms might be derived from EXAFS by the power dependence of ossilcation amplitude50. Due to this fact, we carried out inverse Fourier transformation to check the coordination atoms, displaying that the primary coordination shell consists of Co-S and Co-O (Supplementary Fig. 10 d–f). Wavelet-transformed EXAFS (WT-EXAFS) reveals that the utmost within the area of the primary coordination shell is overlapped by Co-O scattering and Co-S scattering, additional confirming that Co is coordinated by each S and O in CoS2-10 (Fig. 2g). The native construction of Co is quantitatively studied by the least-squares becoming of EXAFS, displaying that Co atoms are coordinated by 4 S atoms at 2.27 Å and two O atoms at 2.00 Å (Supplementary Fig. 11 and Supplementary Desk 2). Due to this fact, we speculate that the CoS2 is reconstructed to cobalt-oxysulfide, as proven in Fig. 2h.
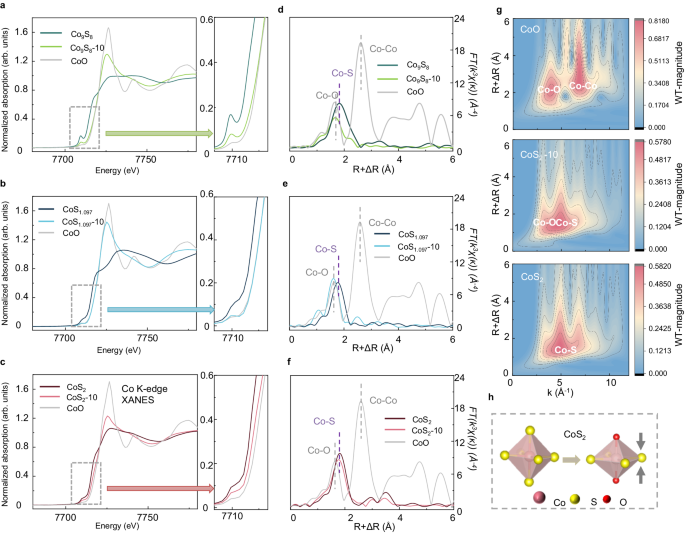
Co Ok-edge XANES of a Co9S8, b CoS1.097, and c CoS2 earlier than and after 10 cycles. Co Ok-edge FT-EXAFS of d Co9S8, e CoS1.097, and f CoS2 earlier than and after 10 cycles. g WT-EXAFS of CoO, CoS2-10 and CoS2. h Schematic of structural evolution of CoS2 throughout biking. Supply knowledge are supplied as a Supply Information file.
CO2 discount and evolution response mechanism
The cyclic voltammetry (CV) curves of Co9S8, CoS1.097, CoS2, and CP in CO2 and Ar environment are proven in Supplementary Fig. 12. All batteries exhibit featureless curves within the Ar environment whereas apparent oxidation and discount peaks within the CO2 environment, which signifies the electrochemical inactivity of sulfides and substrate with out CO2 on the vary of two.2~4.7 V. Due to this fact, the reactions throughout discharge and cost primarily depend on CO2 for the cathodes. To realize mechanistic perception into the electrocatalytic course of, ex situ characterizations of merchandise on CoSx electrodes after discharge and cost had been first carried out, as proven in Supplementary Fig. 13, together with SEM (Supplementary Fig. 14), XRD (Fig. 3a, b), Raman spectroscopy (Supplementary Fig. 15). The SEM pictures present that the discharge merchandise are giant and rodlike overlaying the floor of CoS1.097, whereas these on CoS2 and Co9S8 are smaller in Supplementary Fig. 14a–c. The XRD patterns in Fig. 3a present the sign of discharge merchandise might be assigned to Li2CO3 (#PDF22-1141) on CoS1.097. After the cost, though no different peaks are on all cathodes in Fig. 3b, the irregular residues might be simply noticed on CoS1.097 and Co9S8 whereas these on CoS2 will not be observable in Supplementary Fig. 14d–f. These outcomes roughly point out that, compared with CoS1.097 and Co9S8, CoS2 has the next reversibility. Because the discharge merchandise on CoS2 and Co9S8 cannot be clearly recognized, we infer Li2CO3 likely is the discharge product for the three sulfides primarily based on earlier reviews and XRD sample of discharged CoS1.097 in Li-CO2 batteries12,51,52. We additionally carried out Raman spectroscopy in Supplementary Fig. 15 however peaks at 1080 cm−1 similar to vibration of Li2CO3 are weak on discharged CoS2 and Co9S8.
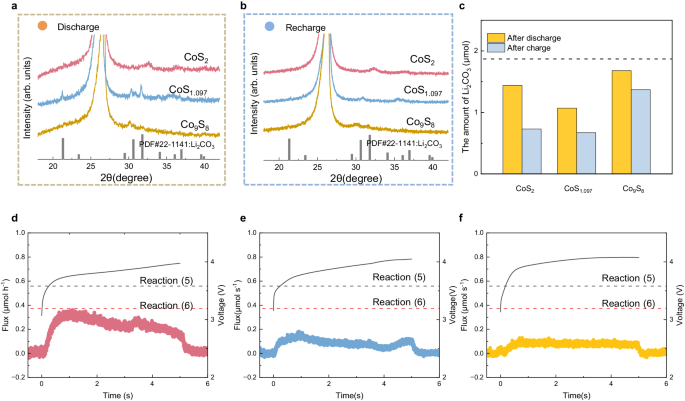
XRD patterns of the three cathodes for a discharging and b charging to 200 μA h cm−2. c The quantity of Li2CO3 formation and residues on three catalysts after discharge and cost. The dashed line is the theoretical worth of Li2CO3 formation after discharge. DEMS outcomes of d CoS2, e CoS1.097, and f Co9S8 throughout cost at a present density of 20 μA cm−2 with a restricted capability of 100 μA h cm−2. The dashed strains are the anticipated flux cost reactions (5) and (6) primarily based on the utilized present density. Supply knowledge are supplied as a Supply Information file.
To confirm our assumption and quantify the reversibility for the three catalysts in Li-CO2 batteries, titration experiments by phosphoric acid are carried out on the catalysts after discharge and cost beneath a present density of 20 μA cm−2 with a restricted capability of 100 μA h cm−2, which in step with electrochemical take a look at (Supplementary Fig. 16)53. As proven in Supplementary Fig. 17, CO2 technology after titrating acid resolution on the discharged catalyst, suggesting carbonates, more than likely Li2CO3 primarily based on the above outcomes, are discharge merchandise on the three catalysts. With exterior customary 1# in Supplementary Fig. 18 and Supplementary Desk 3, the portions of fashioned and residual Li2CO3 throughout discharge and cost on the three cathodes are proven in Fig. 3c and Supplementary Desk 4. By now, the reported doable discharge reactions in Li-CO2 batteries are proven as following reactions (1)–(4)9,54,55,56. The cost to mass of Li2CO3 in all reactions is 2e−/Li2CO3, together with response (2) if Li2C2O4 disproportionates to Li2CO3. For a 2e−/Li2CO3 course of, ~57–89% of the discharge course of goes to the formation of Li2CO3, indicating Li2CO3-related reactions are dominant throughout discharge for the three sulfides. For cost, there are two doable reactions (5) and (6) with Li2CO3 decomposition9,43.
$${4{{{{{rm{Li}}}}}}}^{+}+{4{{{{{rm{e}}}}}}}^{-}+{3{{{{{rm{CO}}}}}}}_{2}to {2{{{{{rm{Li}}}}}}}_{2}{{{{{{rm{CO}}}}}}}_{3}+{{{{{rm{C}}}}}}$$
(1)
$${4{{{{{rm{Li}}}}}}}^{+}+{4{{{{{rm{e}}}}}}}^{-}+{2{{{{{rm{CO}}}}}}}_{2}to {2{{{{{rm{Li}}}}}}}_{2}{{{{{{rm{C}}}}}}}_{2}{{{{{{rm{O}}}}}}}_{4}$$
(2)
$${2{{{{{rm{Li}}}}}}}^{+}+{2{{{{{rm{e}}}}}}}^{-}++ {2{{{{{rm{CO}}}}}}}_{2}to {{{{{{rm{Li}}}}}}}_{2}{{{{{{rm{CO}}}}}}}_{3}+{{{{{rm{CO}}}}}}$$
(3)
$${2{{{{{rm{Li}}}}}}}^{+}+{2{{{{{rm{e}}}}}}}^{-}+{{{{{{rm{CO}}}}}}}_{2}+{{{{{rm{O}}}}}}to {{{{{{rm{Li}}}}}}}_{2}{{{{{{rm{CO}}}}}}}_{3}$$
(4)
$${2{{{{{rm{Li}}}}}}}_{2}{{{{{{rm{CO}}}}}}}_{3}+{{{{{rm{C}}}}}}to {4{{{{{rm{Li}}}}}}}^{+}+{3{{{{{rm{CO}}}}}}}_{2}+{4{{{{{rm{e}}}}}}}^{-}$$
(5)
$${{{{{{rm{Li}}}}}}}_{2}{{{{{{rm{CO}}}}}}}_{3}to {2{{{{{rm{Li}}}}}}}^{+}+{{{{{{rm{CO}}}}}}}_{2}+1/{2{{{{{rm{O}}}}}}}_{2}/{{{{{rm{O}}}}}}+{2{{{{{rm{e}}}}}}}^{-}$$
(6)
The ratio of Li2CO3 and CO2 is ~0.67 for response (5) and 1 for response (6). In situ differential electrochemical mass spectrometry (DEMS) evaluation was carried out to calculate CO2 technology through the cost on the three sulfides. Determine 3d–f and S19 present that solely CO2 (m/z = 44) technology might be noticed on all cathodes and the quantity of CoS2 is way greater than that on Co9S8 and CoS1.097. Because the numerical outcomes are summarized in Supplementary Desk 5, the ratio of Li2CO3 to CO2 of CoS1.097 is 0.85, near 1, indicating that response (6) might largely occur throughout cost. Though no sign of O2 (m/z = 32) has been noticed, oxygen species technology is often doable and threatens the catalyst’s durability43. Consequently, CoS1.097 is oxidized to CoO throughout biking and impacts its electrochemical efficiency. The ratio of Co9S8 and CoS2 is ~0.76 and ~0.56 respectively, near that of the response (5). Nevertheless, the conversion effectivity of Li2CO3 on Co9S8 is barely 18.4% a lot decrease than the opposite cathodes, indicating oxidation reactions primarily occurred to produce capability. The XAS ends in Fig. 2 present that the valence states of Co in three sulfides keep +II after 10 cycles, excluding the opportunity of Co contribution to the cost capability. As an alternative, the sulfur oxidation could also be accountable for the cost capability of Co9S8, because the decreased depth of Co-S binding in Supplementary Fig. 8a. We can also’t exclude the opportunity of electrolyte decomposition that provides the capability and oxidizes the catalysts. Against this, the upper cost effectivity of CoS2 advantages its reconstruction to Co-S4-O2 as an alternative of full oxidation. We additional titrated cathodes after the ninth cost, tenth discharge, and tenth cost to research the battery response on the reconstructed CoS2. As proven in Supplementary Fig. 20, there was little CO2 technology after titrating the cathodes after the ninth and tenth cost, whereas an apparent CO2 technology peak might be noticed on the cathode after the tenth discharge, suggesting that almost all Li2CO3 might be decomposed after cost throughout biking. For the reason that measurement values of CO2 technology on cathodes after the ninth and tenth cost are lower than 5% of that on the cathode after the tenth discharge, we approximate the quantity of Li2CO3 on the cathode after the tenth discharge because the portions of Li2CO3 formation and decomposition within the tenth cycle. Based mostly on exterior customary 2#, the quantity of Li2CO3 formation is ~1.15 μmol, suggesting ~60% cost goes to type Li2CO3 throughout discharge within the tenth cycle (Supplementary Fig. 21 and Supplementary Desk 3). These outcomes exhibit that Li2CO3 stays the principle discharge product and might be virtually fully decomposed through the cost on the reconstructed CoS2 in biking.
DFT calculations and dialogue
DFT calculations had been carried out to elucidate the connection between sulfide construction and exercise in Li-CO2 batteries. Based mostly on our experimental outcomes, we constructed 4 substrates, together with three pre-catalysts Co9S8, CoS1.097, CoS2 and oxygen partially substituted CoS2 after biking (denoted as O-CoS2 within the latter dialogue) in Supplementary Fig. 22. The adsorption energies of CO2, Li, and Li2CO3 had been first calculated to evaluate the interplay between substrates and reactants throughout cost and discharge in Li-CO2 batteries as proven in Supplementary Figs. 23–2618,57,58. As summarized in Fig. 4a and Supplementary Desk 6, the adsorption strengths of CO2, Li, and Li2CO3 on CoS2 are greater than these on different pre-catalysts, besides that the adsorption power of CO2 is little weaker than that on Co9S8. Notably, a extra unfavourable worth of adsorption energies on O-CoS2 than CoS2 signifies partial oxygen substitution successfully will increase the adsorption strengths. The digital modulation by O substitution is revealed in Supplementary Fig. 27 and Supplementary Desk 7, displaying the cost redistribution on neighboring cobalt atoms. A extra constructive area on O-CoS2 than CoS2 means that O substitution will increase the native polarity and interplay with adsorbed species proven in Fig. 4b. Apart from, the d-band middle of Co shifts to the next power degree associated to the Fermi degree on account of oxygen substitution, additionally corresponding with elevated adsorption power of O-CoS2 (Fig. 4c).
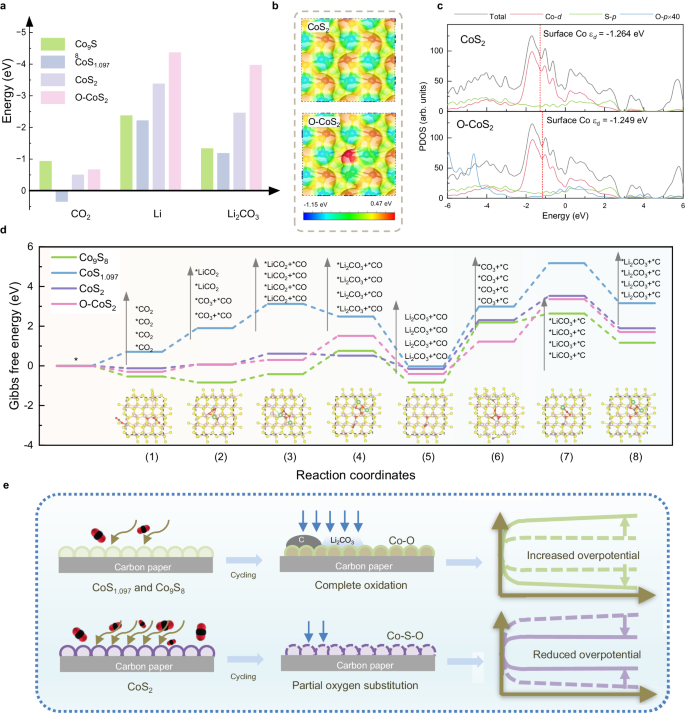
a The CO2, Li, and Li2CO3 adsorption energies on Co9S8, CoS1.097, CoS2 and O-CoS2. b Floor electrostatic potential diagrams of CoS2 (up) and O-CoS2 (down). c The projected density of states (PDOS) of CoS2 (up) and O-CoS2 (down); the inset pink dotted line is the d-band middle. d Gibbs free power diagram of response pathways in Li-CO2 batteries at U = 2.85 V on the 4 catalysts. The inset reveals the highest views of adsorption techniques on O-CoS2. e Illustration of the connection between structural evolution and exercise of CoSx in Li-CO2 batteries. Supply knowledge are supplied as a Supply Information file.
The Gibbs free energies at each open circuit (U = 0 V) and equilibrium (U = 2.85 V) potentials for 5 doable pathways on the 4 constructed catalysts to additional decide the response kinetics are proven in Supplementary Figs. 28–32 and Tables S8–11. On the equilibrium potentials, Fig. 4d reveals that *CO3 and *C formation is the rate-determining step for 3 pre-catalysts and CoS2 has the bottom power distinction of this step (2.46 eV) than Co9S8 (3.03 eV) and CoS1.097 (3.02 eV). The speed-determining step of O-CoS2 is modified to step (7) with ΔG (2.14 eV), additionally decrease than that of three pre-catalysts, indicating that partial oxygen substitution additional improves the catalytic potential of CoS2 in Li-CO2 batteries.
Based mostly on our experimental proof and DFT outcomes, the structural evolution and consequent change in electrochemical efficiency are illustrated in Fig. 4d. Co9S8 and CoS1.097 as pre-catalysts present severe parasitic reactions throughout cost in Li-CO2 batteries. Consequently, the 2 catalysts have been absolutely oxidized throughout biking, which passivates the catalytic skills and ends in elevated overpotentials of batteries. Quite the opposite, CoS2 with greater exercise reveals superior electrochemical efficiency and reversibility, of which oxidation is terminated and kinds oxysulfide with Co-S4-O2 motif in Li-CO2 batteries. The partial oxygen substitution will increase the native polarity and the power degree of the d-band middle, which adjusts the adsorption power and thereby reduces the battery overpotential. Briefly, the preliminary properties of sulfides play an important position of their structural evolutions in batteries and thus have an effect on the efficiency of batteries throughout biking. Our discovering additionally demonstrates the lively motifs for reconstructed catalysts, which give insights for understanding the excessive exercise of sulfides and even different transition compounds.
The appliance in Li-CO2 batteries
The excessive exercise of CoS2 and elevated power effectivity owing to partial oxygen substitution are demonstrated within the latter electrochemical take a look at. The CV curves in Supplementary Fig. 33 present a quicker redox response fee between CO2 and C on CoS2, with oxidation and discount currents being greater compared to CoS1.097 and Co9S8. The onset potentials for CO2RR and CO2ER of three cathodes are in contrast similar to 0.25 mA cm−2 proven in Fig. 5a10,14. CoS2 displays extra constructive and decrease potential for CO2RR (2.76 V) and CO2ER (4.27 V) compared to CoS1.097 (2.69/4.39 V) and Co9S8 (2.59/4.42 V), indicating its greater catalytic actions. CoS2 additionally reveals excessive reversibility as greater cost capability (1781.4 μA cm−2) and Coulombic effectivity (CE) at 88.8% within the galvanostatic charge-discharge (GDC) take a look at (Fig. 5b), whereas CoS1.097, Co9S8 and CP have cost capacities of 1370.6, 940.0 and 47.7 μA cm−2 with corresponding CE of 68.8%, 44.2% and seven.2%, respectively.
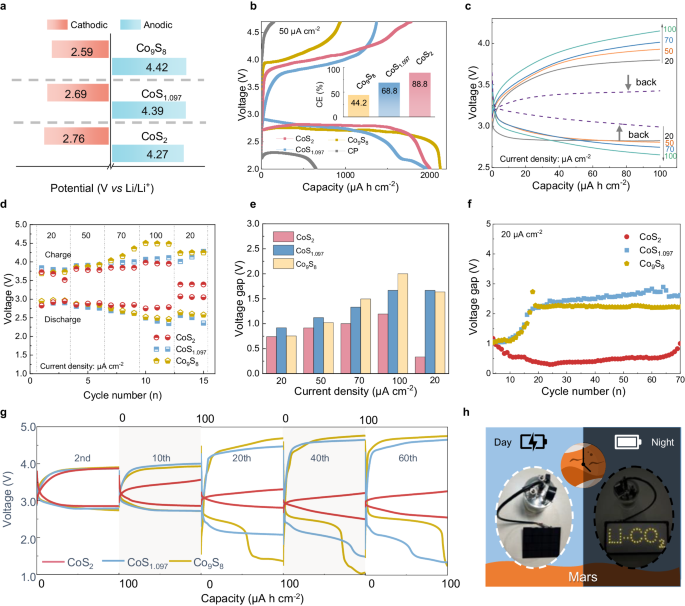
a The onset potentials throughout discharge and cost and b absolutely discharging curves at a fee of fifty μA cm−2 between 2 and 4.7 V (inset is the corresponding CE) for Li-CO2 batteries with the three cathodes. c GDC profiles with a restricted capability of 100 μA h cm−2 at totally different present densities for Li-CO2 battery with the CoS2 cathode. d Discharge and cost voltage and e overpotential at totally different present densities for the three cells. (The worth of overpotential is the common of three cycles with the identical present density). f The voltage hole of the three cells for long-term biking. g GDC profiles of chosen cycles for the three cells. h Picture picture of a solar-powered battery power storage system primarily based on Li-CO2 batteries with CoS2 cathodes at day and evening. Supply knowledge are supplied as a Supply Information file.
Determine 5c–e present the speed efficiency of Li-CO2 batteries with the three cobalt sulfide cathodes. On the present density of 20 μA cm−2, the mid-capacity polarizations of CoS2, CoS1.097, and Co9S8 cathodes are 0.74, 0.91, and 0.75 V, respectively. As the present density elevated to 100 μA cm−2, the overpotentials of the CoS2 cathode are significantly decrease than these of CoS1.097 and Co9S8 cathodes, at just one.19 V, whereas the overpotentials of the latter cathodes are ramped up considerably to 1.67 and a pair of.00 V, respectively. When the present density is reverted to twenty μA cm−2, the overpotential of the CoS2 cathode reduces to 0.33 V even decrease than that of the primary three cycles, in distinction to CoS1.097 (1.67 V) and Co9S8 (1.63 V) cathodes, which exhibit inferior fee skills (Supplementary Fig. 34). The reason for this phenomenon has been elucidated above and the identical in long-term biking. Underneath a present density of 20 μA cm−2, the overpotential of CoS2 is considerably decreased and retains stability in biking as proven in Supplementary Fig. 35a, reducing to 0.43 V after 400 h, which can be higher than different sulfides catalysts (Supplementary Fig. 35b and Supplementary Desk 1). Furthermore, CoS2 additionally maintains a protracted biking lifetime of over 700 h and maintains an overpotential decrease than 1 V till battery failure (Supplementary Fig. 36 and Fig. 5f). In distinction, the overpotential of batteries with CoS1.097 and Co9S8 cathodes exceeds 2 V solely after 20 cycles. These contrasts for 3 cathodes are extra seen within the chosen cycles in Fig. 5g. Determine 5h reveals Li-CO2 cells primarily based on CoS2 electrodes with a solar-powered battery and a light-emitting diode (LED) array at day and evening, which demonstrates its potential in Mars exploration and operation, the place the environment is 96% CO2.
In conclusion, we now have recognized reconstructed motifs and unraveled the structure-activity relations of three cobalt (II) sulfides (CoSx, x = 8/9, 1.097, and a pair of) in Li-CO2 batteries by combining spectroscopy and DFT calculations. We uncover that almost all cobalt atoms in Co9S8 and CoS1.097 coordinate with oxygen atoms after reconstruction, resulting in their deactivation and degradation in battery efficiency. In distinction, partial oxygen substitution with the Co-S4-O2 motif in CoS2 contributes to the cost redistribution on cobalt atoms, thereby enhancing the catalytic potential. Reconstructed CoS2 has a excessive power effectivity (>80%) and superior stability throughout biking with an overpotential of 0.43 V after 400 h in Li-CO2 batteries. Our discovering about lively motifs and digital construction options aids in understanding the excessive exercise of sulfides and different transition compounds catalysts in Li-CO2 batteries. We additionally count on our research can pave the way in which for the event of extremely lively and steady catalysts for metal-gas batteries.








