The competitors mechanism of halogen dopants and Li2Sx in interstitial quantity
The structural chemistry of the state-of-the-art 75Li2S-25P2S5 glassy SSEs, represented by the simplified system Li3PS4, has been properly established. As beforehand reported24, its glass community primarily contains ortho-thiophosphate (PS43−, monomer) and minor portions of hypo-thiodiphosphate (P2S64−, P-P dimer), together with pyro-thiophosphate (P2S74−, dimer) (Fig. S1). Then, to research the adjustments in 75Li2S-25P2S5 glass construction after LiI incorporation, we carried out ab initio molecular dynamic (AIMD) simulations of 75Li2S-25P2S5 glass and LiI-doped 75Li2S-25P2S5 glass by melting and rapid-quenching crystalline Li3PS4 and Li4PS4I (Fig. S2). Our simulation outcomes counsel that the addition of LiI has a minimal impact on the chemistry of the glass community (Figs. S3 and S4). Each 75Li2S-25P2S5 glass and LiI-doped 75Li2S-25P2S5 glass exhibit an identical glass community primarily consisting of PS43−, with small portions of P2S74− and P2S64−. Additionally, our simulation outcomes affirm the perception from the earlier report that the doped LiI doesn’t substitute the sulfur within the PS43−, P2S74− or P2S64−, however dissolves interstitially within the glass network32. In accordance with the regulation of conservation, the formation of P2S64− and P2S74− results in the technology of Li2Sx, which occupies the interstitial positions inside glass community quite than altering its construction. Since each LiI and Li2Sx occupy interstitial websites, the Vint stuffed by Li2Sx hinders the extra dissolution of LiI. Ideally, in a LiI-doped 75Li2S-25P2S5 glass with the simplified system Li3PS4-xLiI, a big Vint could be obtainable for the dissolution of LiI if the glass community consisted solely of PS43−. Nevertheless, facet reactions occurring through the precise preparation course of inevitably result in the formation of P2S64− and P2S74−, leading to a hybrid community comprising each monomers and dimers. This ends in a comparatively smaller Vint in comparison with that of a PS43−-based glass community for the incorporation of LiI, as Li2Sx occupies a sure quantity (Fig. 1b).
Design of high-performance glassy sulfides
Recognizing the competitors of interstitial positions between LiI and Li2Sx, it’s obvious that decreasing the relative quantity of Li2Sx within the glass system will enhance the obtainable Vint, thereby facilitating additional dissolution of LiI. The formation of Li2Sx species stems from the formation chemistry of P2S64− and P2S74− through the preparation course of, necessitating the tracing of such reactions (Fig. 1c). The formation of P2S64− outcomes from a redox response between P5+ and S2− (Equation (1)). Given the steadiness of P(+V)2S5 molecules at room temperature, it’s believable to deduce that the technology of P(+IV)2S64− is induced by localized overheating ensuing from the ball-milling course of employed in synthesizing glassy electrolytes. Apart from, based on the idea of electron switch, the affect of dipole moments on electron switch reactions is significant34,35. Rising dipole moments of the reactant or aligning the dipole with the route of electron switch can facilitate electron switch reactions. Contemplating this, the numerous dipole moments (5.905 Debye) induced by unsaturated P = S bonds in Li3PS4 molecules could act as inducers for the acceleration of redox reactions (Fig. S5). Relating to the formation of P2S74−, it may be induced by reversible reactions between P2S64− and Li2Sx (Equation (2)), in addition to incomplete reactions between glass formers and glass modifiers. Consequently, a technique geared toward decreasing the ratios of Li2Sx in 75Li2S-25P2S5 programs ought to prioritize lowering the potential for self-redox of Li3PS4 and selling reactions between glass formers and glass modifiers.
$${{{{{{rm{2Li}}}}}}}_{3}{{{{{{rm{PS}}}}}}}_{4}to {{{{{{rm{Li}}}}}}}_{4}{{{{{{rm{P}}}}}}}_{2}{{{{{{rm{S}}}}}}}_{6}+({{{{{rm{x}}}}}}-2)/({{{{{rm{x}}}}}}-1){{{{{{rm{Li}}}}}}}_{2}{{{{{rm{S}}}}}}+1/({{{{{rm{x}}}}}}-1){{{{{{rm{Li}}}}}}}_{2}{{{{{{rm{S}}}}}}}_{{{{{{rm{x}}}}}}}({{{{{rm{redox}}}}}},{{{{{rm{response}}}}}})$$
(1)
$${{{{{{rm{Li}}}}}}}_{4}{{{{{{rm{P}}}}}}}_{2}{{{{{{rm{S}}}}}}}_{6}{+{{{{{rm{Li}}}}}}}_{2}{{{{{{rm{S}}}}}}}_{{{{{{rm{x}}}}}}}leftrightarrow {{{{{{rm{Li}}}}}}}_{4}{{{{{{rm{P}}}}}}}_{2}{{{{{{rm{S}}}}}}}_{7}{+{{{{{rm{Li}}}}}}}_{2}{{{{{{rm{S}}}}}}}_{{{{{{rm{x}}}}}}-1}({{{{{rm{reversible}}}}}})$$
(2)
Accordingly, we suggest a modification of the glass community by incorporating SiS2. Firstly, changing partial P2S5 with SiS2, whose cations possess increased lowest unoccupied molecular orbital (LUMO) values (Fig. 1d) can cut back the general incidence of redox reactions within the glass system. Furthermore, the Li4SiS4 molecule with out unsaturated bonds reveals a considerably decrease dipole second of two.973 Debye in comparison with that of the Li3PS4 molecule. After the incorporation of Li4SiS4, the Li3PS4-Li4SiS4 complicated shows a dipole second of 6.025 Debye, whereas the dipole second of Li3PS4 within the complicated is decreased from 5.905 to 4.760 Debye (Fig. S6). This consequence signifies that the cost compensation ensuing from the coordination of Li+ in Li4SiS4 with the S atom within the P = S bond will assist to lower the dipole second of Li3PS4, which can probably suppress electron switch reactions between P5+ and S2−. Subsequently, as illustrated in Fig. 1e, the interplay between Li3PS4 and Li4SiS4 lowers the general Gibbs free vitality, aiding within the transition from precursors to monomers. Contemplating the above elements, Si-doping will cut back the quantities of P2S64− and P2S74− within the glassy sulfide, thereby decreasing the proportion of Li2Sx to unencumber extra Vint for the incorporation of LiI (Fig. 1f). Moreover, applicable doping ranges of SiS2 within the Li2S-P2S5 system won’t end in a big deterioration of the Li/electrolyte interface, as beforehand reported36,37. Accordingly, a sequence of Si-doped 0.6((75 + 0.5x)Li2S-(25-0.5x)P2S5-xSiS2)-40LiI electrolytes have been synthesized. The investigated programs are thereby denoted as follows: clean (with out SiS2 doping), P:Si = 7 (x = 6.25), P:Si = 6 (x = 7.14), P:Si = 5 (x = 8.33), P:Si = 4 (x = 10).
To validate the proposed design method, atomistic simulations utilizing the AIMD method have been performed. Provided that our AIMD simulation methodology depends on the melt-quenching of crystalline supplies, we choose Li4PS4I, which incorporates the best reported LiI content material amongst crystalline sulfides, for our simulations. Regardless of the LiI content material (33.3 mol%) in Li4PS4I being decrease than that in our experimental samples (40 mol%), the adjustments within the glassy framework construction mirrored through the simulation course of can nonetheless present helpful insights. As illustrated in Fig. 2a, the PS43− polyhedral construction in Li4PS4I glass undergoes important disruption upon equilibration at 600 Okay, which is clear from the sharp peaks positioned at ~2.30 Å and three.60 Å of the P-P pair and the broad peak noticed within the vary of three.70–4.50 Å of the P-S pair within the corresponding g(r) profiles. The presence of P4S74− and P4S64− might be inferred from these observations. Then, the affect of Si doping on I-rich Li4PS4I glass was investigated utilizing Li4.125Si0.125P0.875S4I as a consultant precursor, as proven in Fig. S7. As revealed in Fig. S8, the introduction of SiS44− efficiently stabilizes the preliminary PS43− construction, even at a excessive temperature as much as 2000 Okay. Consequently, the Li4.125Si0.125P0.875S4I glass reveals a predominantly monomeric construction upon equilibration at 600 Okay, as evidenced by the absence of observable dimers within the g(r) profiles (Fig. 2b), thereby validating that Si doping can successfully suppress dimers’ formation. Furthermore, to make sure that the pair distribution outcomes are unbiased of the enter constructions utilized in our modeling, two further validation constructions for each Li4PS4I and Li4.125Si0.125P0.875S4I have been constructed and utilized to the constant molecular simulations, respectively (Fig. S9). Consequently, comparable outcomes are noticed throughout various constructions of the identical supplies (Fig S10). Moreover, whereas the g(r) profile of the Li-I pair signifies full dissociation of LiI in each Li4PS4I and Li4.125Si0.125P0.875S4I glass (Fig. S11), snapshots in Fig. second, e after equilibrium reveal that, in comparison with Li4.125Si0.125P0.875S4I, Li4PS4I reveals important occupancy of Sx2− within the interstitial websites. Contemplating the restricted interstitial house, the presence of those non-framework S atoms will compress I atoms and trigger them to maneuver towards the framework because of the coulombic repulsion. By extracting the radial distribution between S atoms from the framework and I atoms, a development of nearer proximity between sure I atoms and the framework S is noticed in Li4PS4I glass (Fig. 2c), which can hinder additional LiI incorporation.
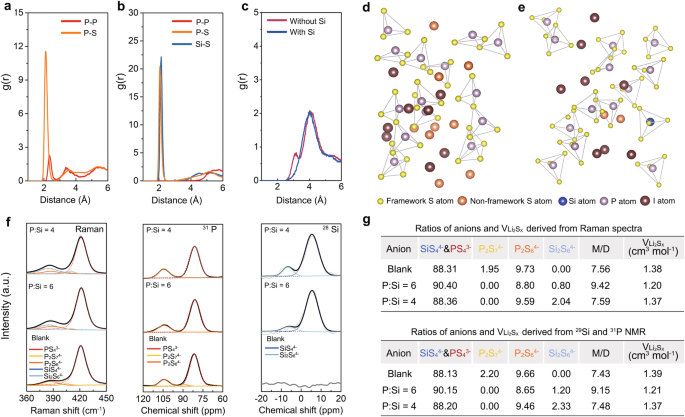
a The g(r) profiles of P-P and P-S pairs within the equilibrium Li4PS4I glass after AIMD simulations. b The g(r) profiles of P-P, P-S, and Si-S pairs within the equilibrium Si-doped Li4.125Si0.125P0.875S4I glass after AIMD simulations. c The g(r) profiles of the framework S-I pair in Li4PS4I glass (with out Si) and Si-doped Li4.125Si0.125P0.875S4I glass (with Si). d, e The snapshot of the equilibrium Li4PS4I glass after AIMD simulations (d) and the snapshot of the equilibrium Si-doped Li4.125Si0.125P0.875S4I glass after AIMD simulations (e). f Raman, 31P and 29Si ss-NMR spectra of the clean, P:Si = 6 and P:Si = 4 electrolytes. g Ratios of anion clusters and minimal VLi2Sx derived from Raman, 31P, and 29Si ss-NMR spectra. (M/D represents the ratio of monomers to dimers).
Moreover, experimental verification of the adjustments in anion clusters as a result of Si doping was performed utilizing Raman spectra mixed with 31P and 29Si solid-state nuclear magnetic resonance (ss-NMR) spectra in clean, P:Si = 6, and P:Si = 4 electrolytes (Fig. 2f). Primarily based on prior Raman/ss-NMR research on Si and P-based sulfides24,38, three peaks similar to PS43− (421.0 cm−1 in Raman, 83.1 ppm in 31P NMR), P2S64−(386.9 cm−1 in Raman, 105.0 ppm in 31P NMR) and P2S74−(404.5 cm−1 in Raman, 88.9 ppm in 31P NMR) are noticed within the clean electrolyte in each Raman and ss-NMR spectra. Upon partial substitution of P5+ by Si4+, the P2S74− peak regularly disappears accompanied by the emergence of SiS44− (385.0 cm−1 in Raman, 0.5 ppm in 29Si NMR) and Si2S64− (410 cm−1, −11.7 ppm in 29Si NMR)29,38. Then, ratios of anion clusters in several electrolytes have been derived utilizing the relative peak space in Raman spectra and the quantitative evaluation of 31P and 29Si ss-NMR spectra within the a part of strategies. As demonstrated in Fig. 2g, Si-doping successfully lowers the ratio of P2S74− and P2S64−, as anticipated by our technique. Nevertheless, extra Si incorporation (P:Si = 4) promotes appreciable Si2S64− formation, which in flip decreases the ratio of monomers. Due to this fact, the utmost ratio of PS43− and SiS44− monomers is reached when the P to Si ratio is 6. By assuming Li2S2 with the smallest quantity amongst lithium polysulfides because the accompanied product of Li4P4S6, and Li2S because the accompanied product of Li4P2S7 and Li4Si2S6, the minimal molar quantity of Li2Sx in every electrolyte might be derived. The minimal molar quantity of Li2Sx within the P:Si = 6 electrolyte is~86% of that within the clean electrolyte, indicating a large amount of Vint is launched to dissolve extra LiI.
Bodily and electrochemical properties of synthesized electrolytes
X-ray diffraction (XRD) evaluation was performed to elucidate the dissolving conduct of LiI in a sequence of synthesized electrolytes. As depicted in Fig. 3a, the XRD spectrum of the clean electrolyte showcases outstanding peaks of LiI at 25.7°, 29.6°, 42.3°, and 50.2°. Upon Si doping, the depth of those sharp LiI peaks diminishes notably within the P:Si = 7 electrolyte and disappears totally within the P:Si = 6 electrolyte. Nevertheless, extreme Si doping fails to facilitate the environment friendly integration of LiI into the glassy community, as evidenced by the restricted launch of interstitial quantity (Vint) within the spectroscopic analyses described above. Consequently, faint LiI peaks are discernible within the XRD spectra of the P:Si = 5 and P:Si = 4 electrolytes. These observations counsel {that a} precipitate-free glassy sulfide with a excessive LiI focus is simply attained on the P:Si = 6 composition.
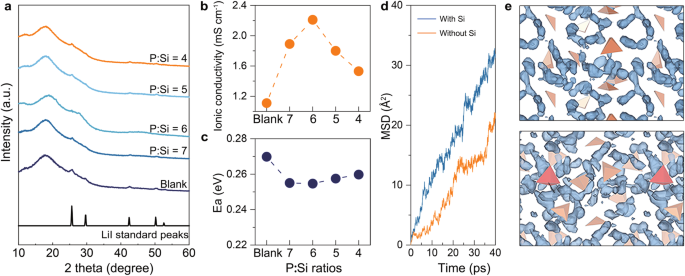
a The XRD spectra of synthesized sequence electrolytes. b The ionic conductivity of synthesized sequence electrolytes. c The Li+ conduction activation vitality of synthesized sequence electrolytes. d The lithium MSD plot for Li4PS4I (orange) and Li4.125Si0.125P0.875S4I (blue) glass at 600 Okay from AIMD simulations. e The Li-ion trajectory (blue) in Li4PS4I (high) and Li4.125Si0.125P0.875S4I (backside) glass at 600 Okay from AIMD simulations. The orange tetrahedra represents PS43−, the purple tetrahedra represents SiS44−.
The ensuing P:Si = 6 electrolyte reveals a glass-transition temperature (Tg) of 132 °C and a crystallization temperature of 206 °C, as decided by differential scanning calorimetry (DSC) evaluation (Fig. S12). Moreover, the pellet of the P:Si = 6 electrolyte shows a flat and compact floor following cold-pressing (Fig. S13a). By hot-pressing the pellet at Tg in a single day, a extremely densified pellet with a relative density of ~100% was obtained (Fig. S13b). Plus, the XRD results of the hot-pressing pellet signifies that the glass nature of the pattern stays unaltered after hot-pressing (Fig. S14). Then, the relative density of the cold-pressed P:Si = 6 pellet is decided to be 93.6% by evaluating the density of the cold-pressed and extremely densified pellet (Desk S1). Moreover, vitality dispersive spectroscopy (EDS) mapping outcomes reveal a homogenous distribution of components inside each the micro-particle and the pellet of the P:Si = 6 electrolyte (Figs. S15–S16).
The ionic conductivity and Li+ conduction activation vitality (Ea) of the synthesized electrolytes have been evaluated utilizing temperature-dependent electrochemical impedance spectroscopy (EIS) evaluation. The related knowledge, together with weight, thickness, Nyquist plots, and Arrhenius plots, are offered in Desk S2, Fig. S17, and Fig. S18, respectively. As depicted in Fig. 3b, the optimum ionic conductivity worth of two.21 mS cm−1 is achieved at a P:Si ratio of 6, which represents a twofold enhance in comparison with the clean electrolyte and ranks one of many highest amongst reported glassy sulfides (Desk S3). Moreover, on the P:Si ratio of 6 (Fig. 3c), the bottom Ea worth of 0.253 eV can be attained. On one hand, the incorporation of LiI into the glassy P:Si = 6 electrolyte, as an alternative of present as an impurity part, results in a rise within the Li+ focus, thereby enhancing Li transport kinetics. Alternatively, Si doping itself may additionally considerably contribute to facilitating Li+ conduction. As demonstrated in Fig. 3d, after leisure for 40 ps at 600 Okay, the lithium imply sq. displacement (MSD) worth of Li4.125Si0.125P0.875S4I glass is increased than that of Li4PS4I glass, indicating a extra unrestrained surroundings for Li+ movement enabled by Si doping. Apart from, in contrast with Li4PS4I glass, Li4.125Si0.125P0.875S4I glass not solely reveals an total enchancment in Li+ transport dynamics but in addition presents a very promoted Li+ transport in proximity to SiS4 tetrahedra positions, which can be attributed to the elevated Li+ focus close to SiS4 tetrahedra and entropy-stabilized impact of Si doping (Fig. 3e)39. Moreover, the Li+ diffusion kinetics in Li4PS4I, Li4.125Si0.125P0.875S4I, and Li3.125Si0.125P0.875S4 have been in contrast (Fig. S19). It may be noticed that solely Si or LiI doping is inadequate to raise lithium transport dynamics to the extent exhibited by Li4.125Si0.125P0.875S4I glass with each Si and LiI doping. Due to this fact, it may be concluded that each the results of LiI dissolution and Si incorporation on the Li+ transport kinetics must be taken accounts to elucidate the numerous enhancement of ionic conductivity in our optimum glass. Plus, to keep away from the affect of the precise enter configuration on the outcomes of diffusion charges, the Li+ diffusion kinetics in two further constructions of Li4PS4I and Li4.125Si0.125P0.875S4I glass have been additionally calculated. As Fig S20 illustrates, distinct constructions of the an identical materials exhibit analogous Li+ diffusion kinetics, which confirms the reliability of this theoretical discovering.
The Li intrusion suppression functionality of the synthesized electrolytes was assessed through CCD assessments with a present step dimension of 0.1 mA cm−2 and a constant charging/discharging length of 1 h for every step (Fig. S21). As demonstrated in Fig. 4a, the clean electrolyte with important LiI impurity demonstrates a CCD worth of 0.6 mA cm−2, which is akin to beforehand reported values33. With Si doping, the CCD worth reveals a development of enhance adopted by a lower, peaking on the P:Si ratio of 6, the place it reaches the best worth of 1.2 mA cm−2. Remarkably, the CCD worth of the P:Si = 6 electrolyte stands out as extremely aggressive amongst glassy sulfides (Desk S4). Moreover, to comprehensively assess the dendrite suppression potential of the P:Si = 6 electrolyte, galvanostatic charging/discharging check with mounted capability was performed to eradicate the results of void-formation40,41. The P:Si = 6 electrolyte reveals secure performance even beneath present density escalation as much as 4.0 mA cm−2 when the discharging/charging capability is maintained at 0.1 mAh cm−2, indicating its potential to face up to demanding operational situations, offered extreme contact loss is averted (Fig. 4b). Nevertheless, it is very important word that the worth derived from this fixed-capacity check could not absolutely characterize the sensible CCD of the SE, the place a bigger capability (>1.0 mAh cm−2) is required. Lengthy-term galvanostatic discharging/charging assessments have been performed on Li symmetric cells utilizing the P:Si = 6 electrolyte to reveal the biking stability. At a low present density of 0.1 mA cm−2, the sluggish Li plating/stripping course of ensures immediate creeping of Li and sustains the contact between Li and the P:Si = 6 electrolyte. Consequently, the Li symmetric cell utilizing the P:Si = 6 electrolyte displayed secure biking conduct for 8000 h even at a excessive cut-off capability of 0.5 mAh cm−2 (Fig. 4c). Moreover, at a low cut-off capability of 0.1 mAh cm−2, when the quantity change of Li steel is insignificant, the symmetric cell with the P:Si = 6 electrolyte demonstrates secure operation for over 1500 h at a present density of 1.0 mA cm−2 (Fig. 4d). The biking stability exhibited by Li symmetric cells using the P:Si = 6 electrolyte can be extremely aggressive when in comparison with beforehand reported situations using glassy electrolytes because the interlayer (Desk S5).
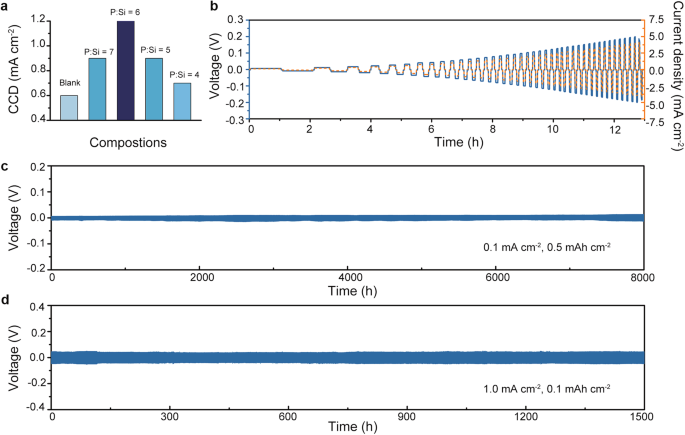
a The abstract of CCD of synthesized electrolytes. b Galvanostatic Li plating/stripping profiles of the Li symmetric cell using the P:Si = 6 electrolyte at step-increased present densities. The capability is mounted to be 0.1 mAh cm−2. c Galvanostatic Li plating/stripping profiles of the Li symmetric cell using the P:Si = 6 electrolyte at a present density of 0.1 mA cm−2 and a cut-off capability of 0.5 mAh cm−2. d Galvanostatic Li plating/stripping profiles of the Li symmetric cell using the P:Si = 6 electrolyte at a present density of 1.0 mA cm−2 and a cut-off capability of 0.1 mAh cm−2.
The comparability between glassy P:Si = 6 and state-of-the-art sulfides
The state-of-the-art argyrodite sulfide Li6PS5Cl and the 75Li2S-25P2S5 glassy electrolyte, generally used as interlayers for Li steel, have been chosen as comparability teams to additional consider the efficiency of the P:Si = 6 electrolyte. In accordance with the EIS spectra at 25 °C (Fig. S22), the ionic conductivity of 75Li2S-25P2S5 and Li6PS5Cl is 0.46 mS cm−1 and a couple of.04 mS cm−1, respectively, that are according to the earlier research42,43 and decrease than that of the P:Si = 6 electrolyte. Moreover, digital conductivity values of 75Li2S-25P2S5, Li6PS5Cl and the P:Si = 6 electrolyte have been measured. Among the many three electrolytes, the P:Si = 6 electrolyte shows the bottom digital conductivity of 1.50 × 10−9 S cm−1, whereas 75Li2S-25P2S5 and Li6PS5Cl exhibit digital conductivity values of two.60 × 10−9 S cm−1 and 4.12 × 10−9 S cm−1, respectively (Fig. S23). Plus, the micro-morphology of Li6PS5Cl, 75Li2S-25P2S5 and the P:Si = 6 electrolyte is represented in Fig. S24. Intimately, the polycrystalline Li6PS5Cl presents apparent edges and voids created by non-dense crystal-crystal stacking whereas 75Li2S-25P2S5 glass with long-range dysfunction construction shows a denser stacking with out noticeable edges and voids. Particularly, a cobblestone construction with a flat and clean grain floor is noticed within the I-rich P:Si = 6 glass with a gentle nature, indicating ignorable interfacial vitality between grains. Furtherly, the cold-pressed Li6PS5Cl pellet reveals a comparatively low relative density of 84.8% attributed to in depth grain boundaries. As compared, the relative densities of the glass electrolyte 75Li2S-25P2S5 and P:Si = 6 are 91.0% and 93.6%, respectively, each surpassing that of Li6PS5Cl (Desk S6). Notably, the P:Si = 6 electrolyte demonstrates the next relative density than 75Li2S-25P2S5. That is primarily attributed to the halogen dopant LiI, which successfully reduces the elastic modulus of sulfide electrolytes, thereby enhancing the relative density of cold-pressed electrolyte pellets10.
Then, Li stripping/plating Coulombic effectivity (CE) values in three SSEs have been evaluated through the use of the Li || present collector half cell. As a result of potential reactivity of copper with sulfide electrolytes, stainless-steel (SS) is utilized as the present collector materials as an alternative of copper. To make sure enough contact between the SS and electrolytes, a modest present density of 0.1 mA cm−2 and a cut-off capability of 0.1 mAh cm−2 have been utilized for Li stripping/plating on the SS. Within the first cycle, the best preliminary CE worth of 86.08% is achieved within the P:Si = 6 electrolyte whereas Li6PS5Cl and 75Li2S-25P2S5 reveal decrease preliminary CE values of 76.50% and 72.62%, respectively (Fig. 5a). Moreover, because the cycle progresses, the P:Si = 6 electrolyte demonstrates the best common CE worth of 86.68%, surpassing that of Li6PS5Cl (75.75%) and 75Li2S-25P2S5 (76.28%). The corresponding voltage-capacity profiles of Li || SS cells are offered in Fig.S25.
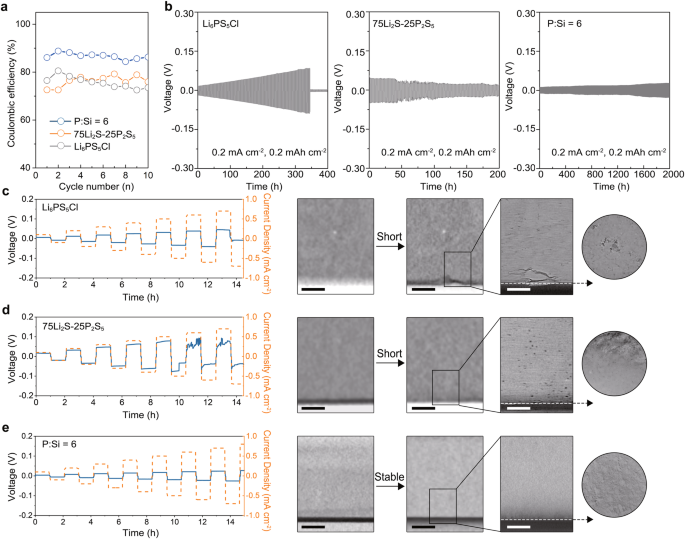
a The CE profiles of three electrolytes. b Lengthy-time galvanostatic biking profiles of three electrolytes. c–e The profiles of the CCD check on Li6PS5Cl-based Li symmetric cell and the in-situ X-ray CT morphology of the Li6PS5Cl/Li interface earlier than and after the CCD check (c). The profiles of the CCD check on 75Li2S-25P2S5-based Li symmetric cell and the in-situ X-ray CT morphology of the 75Li2S-25P2S5/Li interface earlier than and after the CCD check (d). The profiles of the CCD check on P:Si = 6-based Li symmetric cell and the in-situ X-ray CT morphology of the P:Si = 6 /Li interface earlier than and after the CCD check (e). The black scale bar is 250 μm and the white scale bar is 100 μm.
From the above outcomes, it’s evident that 75Li2S-25P2S5 shows a decrease preliminary CE when in comparison with Li6PS5Cl. This discrepancy could also be attributed to a number of potential elements. Firstly, the P2S64− cluster in 75Li2S-25P2S5 possessing a decrease LUMO worth than the PS43− clusters is extra prone to discount by lithium steel (Fig. S26). Apart from, the decrease P content material in Li6PS5Cl results in a diminished presence of Li-P species with sure digital conductivity within the SEI. Nevertheless, the common CE worth of Li6PS5Cl was found to be decrease than that of 75Li2S-25P2S5. This may increasingly stem from the continual infiltration of lithium into the non-dense Li6PS5Cl pellet with cracks throughout electrochemical biking, resulting in elevated electrolyte discount. Relating to the P:Si = 6 electrolyte, its possession of the best preliminary CE and common CE values among the many three electrolytes could also be attributed to 2 elements. Firstly, the P:Si = 6 electrolyte reveals the best relative density among the many three electrolytes, which suppresses the Li intrusion into the electrolyte, thereby decreasing the incidence of recent facet reactions throughout biking. Secondly, the X-ray photoelectron spectroscopy (XPS) evaluation on Li/P:Si = 6 electrolyte interface revealed that, regardless of the presence of Si4+, solely P-cations within the P:Si = 6 electrolyte are diminished to type Li-P species, resulting in the technology of Li2S and LiI (Fig. S27). This aligns properly with the earlier AIMD simulations and spectroscopic characterizations of the Li7SiPS8/Li interface44. In opposition to this backdrop, one key distinction between the P:Si = 6 glass and different two electrolytes lies primarily in its highest halide (LiI) content material and lowest P content material. Upon response with metallic lithium, the P:Si = 6 electrolyte types the SEI characterised by a big quantity of LiI and minimal Li-P species. In comparison with Li-P species with a bandgap of 0.70–1.76 eV, LiI possesses a broader bandgap of 4.37 eV (Fig. S28), which can successfully cut back the general digital conductivity of the interphase, thereby suppressing additional facet reactions.
Subsequently, long-time galvanostatic discharging/charging assessments have been carried out on Li symmetric cells utilizing totally different electrolytes at a present density of 0.2 mA cm−2 and a cut-off capability of 0.2 mAh cm−2. The Li symmetric cells utilizing the P:Si = 6 electrolyte demonstrates secure biking for 2000 h, indicating that the excessive relative density, ample ionic conductivity, and excessive interfacial stability of the P:Si = 6 electrolyte can successfully regulate Li steel intrusion throughout biking. In sharp distinction, brief or micro-short circuits happen inside 400 h for Li symmetric cells utilizing Li6PS5Cl and 75Li2S-25P2S5 electrolytes (Fig. 5b).
An in-situ X-ray computerized tomography (X-ray CT) mixed with ex-situ scanning electron microscopy (SEM) was then carried out to couple the electrochemical behaviors with the morphological evolution. Initially, mildew cells have been assembled and clamped utilizing a normal holder (Fig. S29) earlier than a preliminary X-ray scan was carried out on the pristine Li/electrolytes interface with an 11 μm voxel decision. Galvanostatic discharging/charging assessments with elevated present density have been subsequently carried out on Li symmetric cells utilizing totally different electrolytes. After a complete biking time of 15 h, each gentle and onerous breakdowns are noticed within the Li symmetric cells utilizing 75Li2S-25P2S5 and Li6PS5Cl electrolytes45, respectively, with corresponding CCD values of 0.6 mA cm−2 and 0.7 mA cm−2. In distinction, the Li symmetric cell utilizing the P:Si = 6 electrolyte reveals secure performance all through the check with out sudden voltage drops. A second X-ray scan was then carried out on the cycled Li/electrolyte interface, with a voxel decision just like the primary scan. Comparability of the primary and second scans reveals that the crystalline Li6PS5Cl interlayer reveals cracks with a diameter of 5–10 μm after the cell was brief, whereas the constructions of 75Li2S-25P2S5 and the P:Si = 6 electrolyte stay primarily intact beneath this low decision. A 3rd scan, utilizing a voxel decision of 1.2 μm, was then performed on the circled space to offer extra detailed info. The high-resolution X-ray CT outcomes of the cycled interface point out that the Li6PS5Cl interlayer is considerably deteriorated and the floor of 75Li2S-25P2S5 is corroded by Li steel, accompanied by the technology of voids and pits, whereas an nearly intact Li/electrolyte interface is noticed within the symmetric cell using the P:Si = 6 electrolyte (Fig. 5c–e). Given the invisibility of Li intrusions in X-ray CT assessments, the mildew cells have been disassembled, and SEM was employed to research the incidence of Li intrusions throughout numerous electrolytes. As proven in Fig. S30, violent Li intrusions resulting in the destruction of the pellet construction are noticed on the Li/Li6PS5Cl interface, which is according to prior research15. Conversely, for 75Li2S-25P2S5 with excessive relative density however low ionic conductivity, whereas no notable deformation of the pellet construction is clear, small Li intrusions are noticed at each the interface and throughout the pellet inside, probably contributing to the phenomenon of sentimental breakdown. In distinction, the glassy P:Si = 6 layer with excessive relative density and ample ionic conductivity stays freed from Li intrusions after biking.
Glassy/crystalline composite design
The suppression of Li steel intrusions realized by the P:Si = 6 electrolyte with ample ionic conductivity, low digital conductivity, excessive Li/electrolyte interfacial stability and excessive relative density conjures up us to mix the professionals of the P:Si = 6 electrolyte and high-Cl content material argyrodite Li5.4PS4.4Cl1.6 with excessive ionic conductivity46,47, thereby producing a composite electrolyte reconciling the excessive ionic conductivity, low digital conductivity, excessive Li/electrolyte interfacial stability and excessive relative density to furtherly suppress the Li intrusions. Our design, which includes the incorporation of the P:Si = 6 electrolyte into the high-Cl content material argyrodite Li5.4PS4.4Cl1.6, affords a number of benefits. Firstly, this mixture advantages from superior chemical compatibility between sulfide electrolytes. Prior research have proven that sulfides could react with polymers and halides48,49, however high-Cl content material argyrodite Li5.4PS4.4Cl1.6, being a sulfide, is intrinsically secure towards the P:Si = 6 electrolyte. Secondly, this design permits the upkeep of excessive ionic conductivity. The managed addition of the P:Si = 6 electrolyte with ample ionic conductivity won’t considerably impede Li+ transport, thereby permitting the excessive Li+ diffusion attribute of high-Cl content material argyrodite Li5.4PS4.4Cl1.6 to be preserved. Apart from, the incorporation of the P:Si = 6 electrolyte gives enhanced functionality for suppressing Li steel intrusions. The P:Si = 6 electrolyte successfully fills cracks, flaws, and gaps between grain boundaries of the argyrodite, resulting in elevated relative density50. Furthermore, earlier analysis reveals that high-Cl content material argyrodite reveals good stability towards Li metal30. This research additionally demonstrates the superior stability of the P:Si = 6 electrolyte towards Li steel. Consequently, the composite electrolyte maintains the steadiness towards Li steel to suppress facet reactions. Lastly, the incorporation of P:Si = 6 glass with low digital conductivity could cut back the general digital conductivity (Fig. 6a).
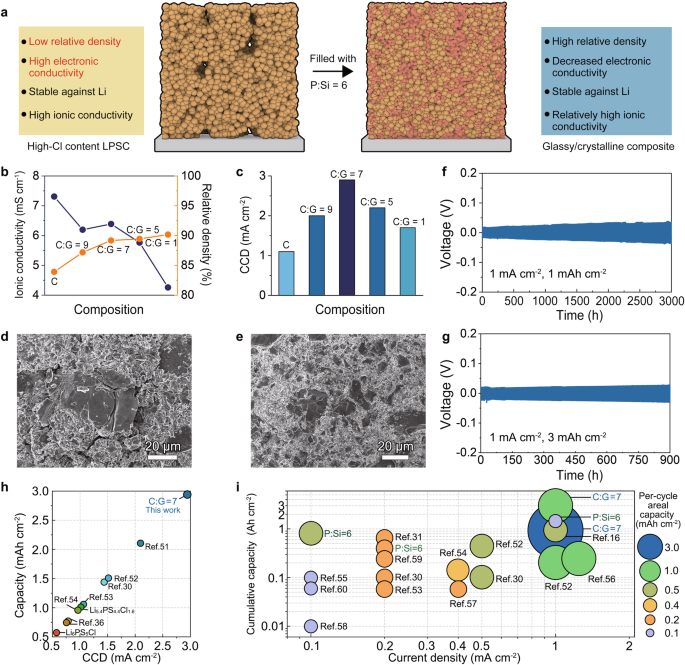
a The schematic diagram of the composite electrolyte design. b The abstract of the relative density and ionic conductivity of the composite electrolyte. c The abstract of CCD of the composite electrolyte. d The floor morphology of the cold-pressed Li5.4PS4.4Cl1.6 electrolyte. e The floor morphology of the cold-pressed C:G = 7 electrolyte. f Galvanostatic Li plating/stripping profiles of the Li symmetric cell using the C:G = 7 electrolyte at a present density of 1 mA cm−2 and a cut-off capability of 1 mAh cm−2. g Galvanostatic Li plating/stripping profiles of the Li symmetric cell using the C:G = 7 electrolyte at a present density of 1 mA cm−2 and a cut-off capability of three mAh cm−2. h The abstract of CCD values for reported sulfide electrolytes coupled with bare-Li steel at 25 °C. See Desk S8 for detailed info of every reference within the determine. i The abstract of biking efficiency of reported bare-Li symmetric cells utilizing sulfide-based electrolytes because the interlayer at 25 °C. See Desk S9 for detailed info of every reference within the determine.
Then, a sequence of composite electrolytes have been ready by ball-milling Li5.4PS4.4Cl1.6 and P:Si = 6 electrolytes at a low pace of 100 rpm. The investigated programs are thereby denoted as C (Li5.4PS4.4Cl1.6), C:G = 9 (10 wt% of P:Si = 6 electrolytes), C:G = 7 (12.5 wt% of P:Si = 6 electrolytes), C:G = 5 (16.7 wt% of P:Si = 6 electrolytes), C:G = 1 (50 wt% of P:Si = 6 electrolytes). XRD part evaluation of electrolytes earlier than and after mixing reveals that the construction of high-Cl content material Li5.4PS4.4Cl1.6 stays unaltered (Fig. S31), and no new species are generated, indicating no response between the P:Si = 6 electrolyte and high-Cl content material Li5.4PS4.4Cl1.6.
The EIS outcomes reveal (Fig. S32) that the preliminary excessive ionic conductivity of the Li5.4PS4.4Cl1.6 electrolyte (7.31 mS cm−1) is maintained at a degree of ~6 mS cm−1 when the load ratio of the P:Si = 6 electrolyte is under 16.7%, as depicted in Fig. 6b. Nevertheless, extreme incorporation of the P:Si = 6 electrolyte results in a notable lower in ionic conductivity to 4.26 mS cm−1 within the C:G = 1 electrolyte. Moreover, when the load ratio of P:Si = 6 electrolyte exceeds 12.5%, the relative density of the electrolyte pellet will increase from 83.9% to over 89% (Fig. 6b). The detailed derivation of relative density of various electrolytes is offered in Desk S7. This phenomenon is confirmed by the outcomes of SEM, which reveals apparent cracks and flaws in native areas of the Li5.4PS4.4Cl1.6 electrolyte (Fig. 6d and Fig. S33a), whereas the pellet of C:G = 7 electrolyte stays crack-free and flat (Fig. 6e and Fig. S33b). Digital conductivity measurements have been then carried out, and the corresponding values for C, C:G = 7, and C:G = 1 electrolytes are decided to be 4.22 × 10−9 S cm−1, 2.54 × 10−9 S cm−1, and a couple of.10 × 10−9 S cm−1, respectively (Fig. S34). This consequence signifies that the incorporation of the P:Si = 6 electrolyte with low digital conductivity reduces the general electron transport within the composite electrolyte.
Subsequently, Li symmetric cells have been assembled utilizing ready composite electrolytes because the interlayer to guage their functionality of suppressing Li intrusions. The CCD check with a present step dimension of 0.1 mA cm−2 was firstly carried out (Fig. S35). Determine 6c illustrates that the Li5.4PS4.4Cl1.6 electrolyte reveals a CCD worth of 1.1 mA cm−2. Incorporating the P:Si = 6 glassy electrolyte into the Li5.4PS4.4Cl1.6 electrolyte considerably enhances the CCD values of the composite, attaining a peak of two.9 mA cm−2 on the composition of C:G = 7 (Fig. 6c). Furthermore, to reveal the prevalence of utilizing I-rich P:Si = 6 electrolyte because the filler, CCD check was carried out on the symmetric cell utilizing Li5.4PS4.4Cl1.6/75Li2S-25P2S5 composite electrolyte, with a weight ratio of 75Li2S-25P2S5 = 12.5%, because the interlayer. The addition of 75Li2S-25P2S5 is discovered to extend the CCD worth to only one.4 mA cm−2 (Fig. S36). Lengthy-time galvanostatic discharging/charging assessments have been carried out on the Li symmetric cells with the C:G = 7 electrolyte. The symmetric cell with the C:G = 7 electrolyte because the interlayer demonstrates secure biking for over 3000 h at a present density of 1 mA cm−2 and a cut-off capability of 1 mAh cm−2 (Fig. 6f). In sharp distinction, the symmetric cell utilizing Li5.4PS4.4Cl1.6 electrolyte because the interlayer can solely operate for six h (Fig. S37). Following the analysis protocol proposed by Wang et al.45, the Li symmetric cell utilizing the C:G = 7 electrolyte was examined at an areal capability of three mAh cm−2 and a present density of 1 mA cm−2. Notably, secure biking for over 900 h is noticed, indicating the sensible utility potential of glassy/crystalline composite electrolytes in excessive energy-density ASSLMBs (Fig. 6g). It’s noteworthy that the CCD worth and biking performances of the C:G = 7 electrolyte in Li symmetric cells stand out prominently amongst sulfide electrolytes, as Fig. 6h, i illustrates16,30,31,36,51,52,53,54,55,56,57,58,59,60.
To additional elucidate the electrochemical efficiency of the C:G = 7 electrolyte, ASSLMBs have been constructed by pairing a high-voltage NCM83125 cathode with a 100 μm Li foil (Fig. 7a). The electrochemical performances of the ASSLMBs have been evaluated by galvanostatic discharging/charging assessments utilizing Li5.4PS4.4Cl1.6 or C:G = 7 electrolytes because the interlayer. The ASSLMBs utilizing Li5.4PS4.4Cl1.6 and the C:G = 7 electrolyte exhibit comparable preliminary discharging capability values of 192.22 mAh g−1 and 192.65 mAh g−1, respectively, with corresponding CE of 82.84% and 83.53% when activated at 0.1 C (0.125 mA cm−2). After 3 cycles of activation, the cells have been then cycled at 0.2 C (0.25 mA cm−2). The discharging capability of the Li | Li5.4PS4.4Cl1.6 | NCM83125 full cell reveals a continuing lower, which is attributed to elevated battery resistance attributable to facet reactions between intruded Li and Li5.4PS4.4Cl1.6. A brief circuit happens across the thirtieth cycle, as indicated by fluctuations within the charging profiles (Fig. S38) and a big lower in CE. In distinction, the complete cell utilizing the C:G = 7 electrolyte demonstrates secure discharging/charging behaviors throughout biking (Fig. 7b, c). Moreover. when cycled at 0.25 mA cm−2, the Li | C:G = 7 | NCM83125 full cell delivers an preliminary discharging capability of 172.12 mAh g−1, which is increased than that of the Li | Li5.4PS4.4Cl1.6 | NCM83125 full cell (163.23 mAh g−1). On the five hundredth cycle, the complete cell utilizing the C:G = 7 electrolyte maintains a discharging capability of 141.98 mAh g−1 with a capability retention of 82.4% (Fig. 7b), which is a extremely aggressive consequence in comparison with reported sulfide-based ASSLMBs (Fig. 7d)30,52,61,62,63,64,65,66,67,68,69.
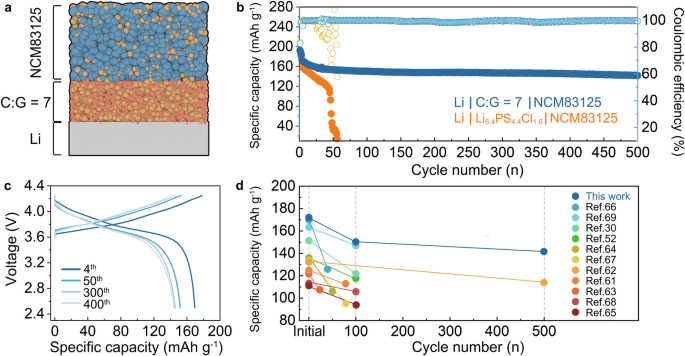
a The configuration of the Li | C:G = 7 | NCM83125 full cell. b Biking performances of Li | Li5.4PS4.4Cl1.6 | NCM83125 and Li | C:G = 7 | NCM83125 full cells at 0.25 mA cm−2. A present density of 0.125 mA cm−2 was utilized to activate the complete cell for the primary 3 cycles. c The voltage profiles of Li | C:G = 7 | NCM83125 full cell at totally different cycles. d The abstract of biking efficiency of reported high-voltage ASSLMBs. See Desk S10 for detailed reference of every level.
In abstract, by deciphering the formation chemistry of the glass community, we suggest Vint as a significant consider figuring out the halogen dopant solubility in sulfide glass electrolytes. Primarily based on the insights of theoretical calculations on the glass forming course of, we launch the Vint within the state-of-the-art 75Li2S-P2S5 glass by establishing a monomer-rich glass community to dissolve extra LiI dopants. The ensuing glass with a excessive ionic conductivity of two.21 × 10−3 S cm−1, a excessive relative density of 93.6%, and a low digital conductivity of 1.50 × 10−9 S cm−1 capabilities properly in regulating the Li steel suppression. Moreover, this glass surpasses the present main Li6PS5Cl and 75Li2S−25P2S5 when it comes to ionic conductivity, Li intrusions suppressing functionality and interfacial stability with Li. Furthermore, a glassy/crystalline composite design is proposed to mix the professionals of high-Cl content material argyrodite and our optimum glass electrolytes. The composite electrolyte notably suppresses Li steel intrusions. Our composite design achieves secure biking at sensible present densities and areal capacities. Utilizing the composite electrolytes as SSEs, all-solid-state Li | |NCM83125 batteries exhibit a retention of 82.4% of preliminary discharging capability after 500 cycles at a present density of 0.25 mA cm−2. This research gives insights into the dissolution chemistry of halogen dopants in glass and a sensible roadmap for creating progressive electrolyte designs. Moreover, the insights from this research may apply past battery expertise, providing helpful methods for the design and optimization of practical supplies with improved properties through tailor-made dopant incorporation.








